The Penrose Inquiry recommendation
Short-life working group report on the Penrose Inquiry recommendation on infection of people with Hepatitis C from NHS treatment.
Appendix 4: Testing of Scottish National Blood Transfusion Service donor archive samples collected and stored prior to the implementation of routine anti- HCV testing
1 Introduction
The Penrose Inquiry Final Report (March 2015) made a single recommendation: 'That the Scottish Government takes all reasonable steps to offer an HCV test to everyone in Scotland who had a blood transfusion before September 1991 and who has not been screened for HCV'.
In evaluating options to address this recommendation, SNBTS has been asked to assess the feasibility of testing archived donor samples from 1984 to 1991 for the presence of HCV. Although the reference period for the Inquiry was 1 st January 1974 to 1 st September 1991 there are no archived samples from prior to 1984. Routine testing of all donations for anti- HCV antibodies commenced as a mandatory requirement from September 1991.
Since 1984 SNBTS has, where possible, retained a small sample from every blood donation which are stored frozen, off site, in a warehouse. The main purpose of the archived samples is to support the investigation into infections or adverse reactions in patients where the transfusion of blood components may be implicated. The testing of archive samples from implicated donors can help confirm whether the transfused blood components were the source of the infection or adverse reaction. It is unusual for a Blood Service to retain a sample archive of this duration, there is no legal requirement to do so and other Blood Services only retain donor samples for up to 3 years. Around 10 years ago SNBTS considered discarding the older samples in the archive due to concerns over their identification, provenance and key data linkages, however this was put on hold because of the genesis of the Inquiry.
In conducting this review it was necessary to consider the sample archive over two different periods, the first being all the samples collected and stored between 1984 and 1992 and the second being the National Sample Archive which was established from 1998. The differences between these two archives are significant and are explained in the following sections so that the difficulties and limitations in screening the former can be understood. Archive samples from 1992 to 1998 fall somewhere between the two. The review also explores the availability of SNBTS records from this period, the challenges and costs of performing the actual tests, the availability of hospital blood bank and clinical records and the issues relating to dealing with the outcomes of testing for donors and patients. Finally we make an estimate of the likely feasibility, work and cost involved in retrieving, testing and reconciling the estimated 2 - 2.5 million samples stored from this period.
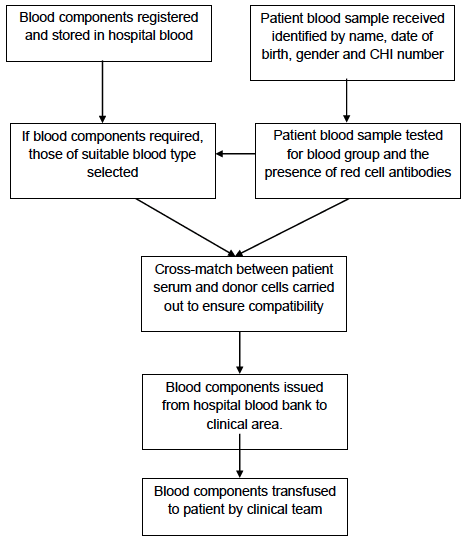
2 Blood Donation, Processing and Testing, Distribution and Transfusion (Appendix 4.1)
We thought it would be helpful to first provide an explanation of the main processes involved from blood donation through to transfusion to a patient and the way in which these are supported across NHS Scotland today through partially-automated testing, positive sample identification (bar codes) and integrated IT systems. These are depicted in the appended high level process maps (Appendices 4.1a - 4.1e). It should be remembered that over the years these systems have replaced manual systems where paper records were the norm, transcriptions errors frequent and timely access poor.
In the current system, there are three essential elements in the tracking of blood components between donors and recipients:
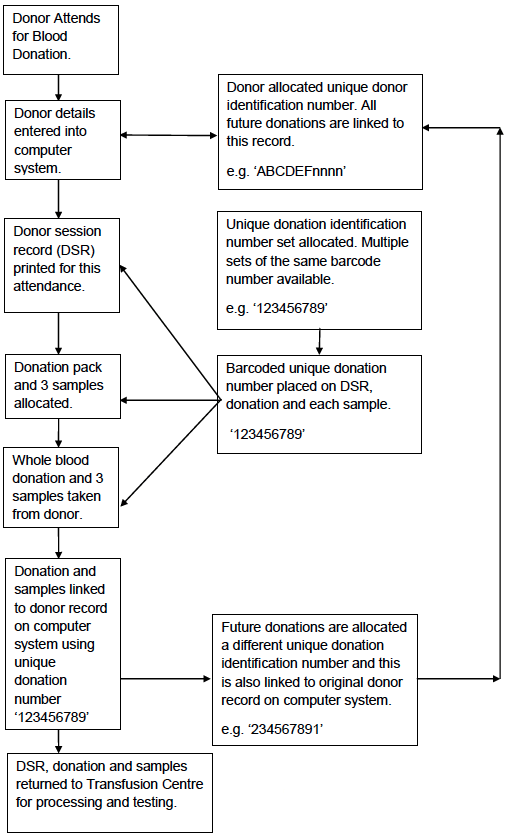
The first is the donor record and the unique identification number created for every donor. This number is used at every attendance and provides assurance that it is the correct donor and record, that has been identified at the collection session (Appendix 4.1a).
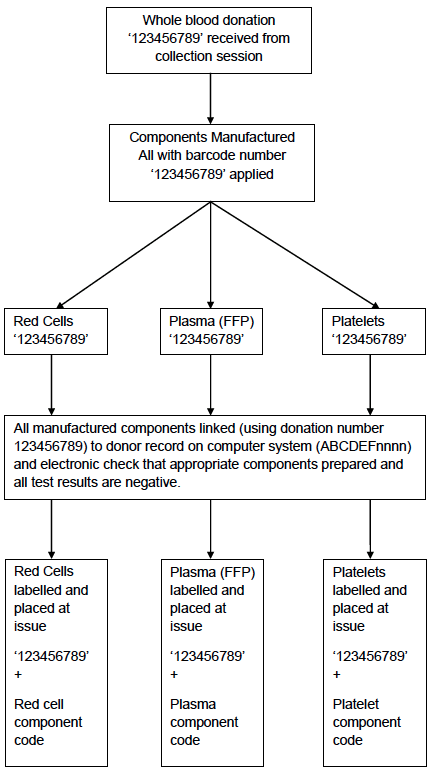
The second is the use of a unique bar-coded donation number which is applied to each donation session record, each donation, the 3 analytical samples taken during blood collection, and to all components manufactured from the donation. This donation number is permanently linked to the donor record. Every time the donor donates a different unique donation identification number is applied to the donation, samples etc and this is again linked to the donor record on the computer system (Appendices 4.1b and 4.1c).
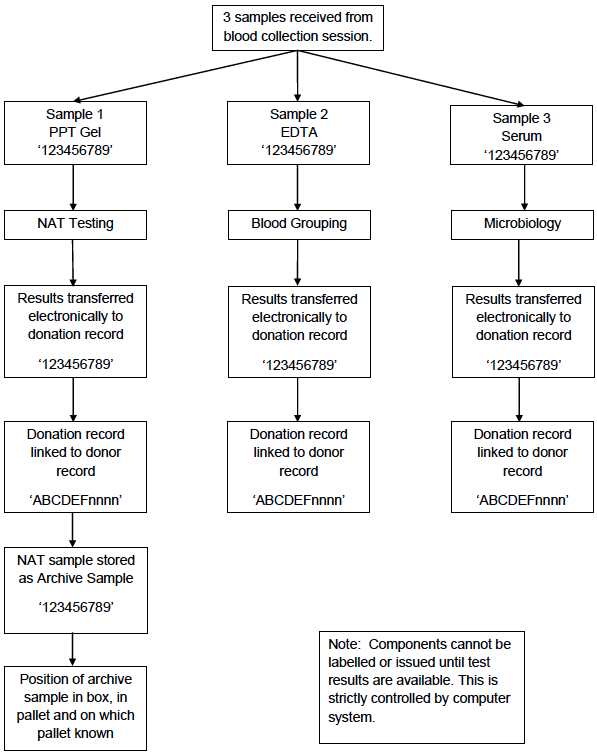
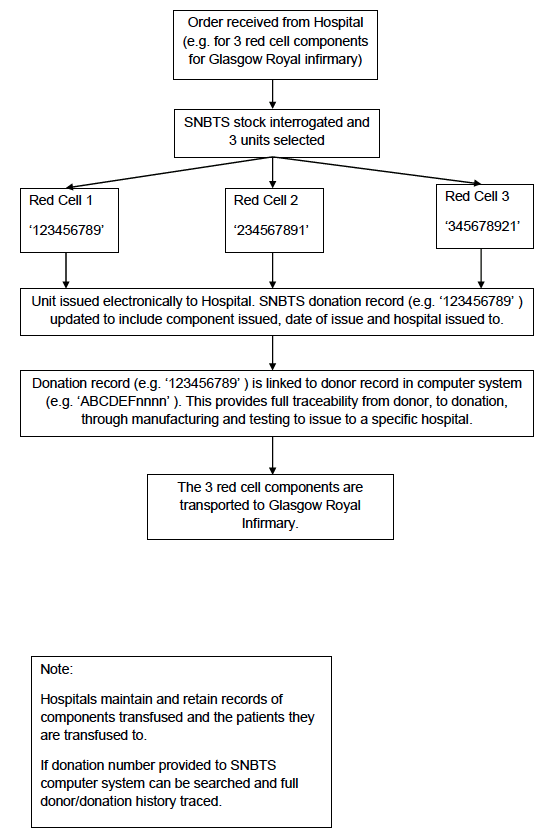
During component manufacture the bar-coded donation identification number is scanned at each processing step and is automatically recorded in the computer system. During testing the bar-coded donation identification number is automatically scanned by the analysers and the results associated with this number electronically transferred into the computer system. The components from the donation cannot be released to hospital blood banks until all the test results are available (and negative). The donation number is also used to track the release of components to hospital blood banks (Appendix 4.1d).
The third is the hospital blood bank itself. The components held are anonymised ( i.e. they don't have the personal details of the donor) but are identified by the unique donation number and component code. Patient blood samples are received from the clinical environment by the hospital blood bank for blood grouping, red cell antibody testing and cross match against potential donations. The patient records are maintained by the hospital blood bank identified by name, gender, date of birth and CHI number. These are reconciled against the donations selected and issued to that patient. The clinical team administering the donation should also record the transfusion in the clinical notes of the patient.
Modern transfusion is therefore highly reliant on robust integrated systems to ensure appropriate records can be maintained for all transactions and that they are readily available and accessible. The use of internationally agreed barcode numbers and the scanning of these numbers at all stages of the process provide a high degree of accuracy and ensure traceability of all elements of the blood supply. SNBTS introduced its first fully integrated computer system to manage donor administration, blood collection, component manufacture, testing, component labelling and issue to hospitals in 1998. Prior to that records were maintained on first generation fragmented computing systems and / or local manual systems (Appendix 4.1e).
3 Archive Samples 1984 - 1991 (Appendix 4.2 and Figure 4.1)
During the period in question SNBTS carried out processing and testing of donations at 5 semi-autonomous Regional Transfusion Centres ( RTCs) in Aberdeen, Dundee, Edinburgh, Glasgow and Inverness. Each RTC retained and stored their own sample archive and a variety of methods and storage containers were employed. Quality, IT and record systems were quite rudimentary when compared to contemporary standards. The methods employed to test donor samples and to prepare each sample archive would not be considered suitable in today's environment.
Archive records for this time period were mainly paper-based, and may now be weak or patchy as evidenced by the poor success rate in tracing pre-1991 samples in previous 'look-back' investigations.
In addition, sample labelling and therefore identification is poor and sample volume low such that automated screening, as is routine now, would not be possible on these samples. Although manual testing is acceptable for small scale testing in investigations it is a very significant undertaking for the large scale screening required for this exercise.
Finally there are concerns around the integrity of the samples after such prolonged storage and confidence in the results obtained would be reduced.
In essence the further one goes back in time within the archive, the more difficult it is to be confident of the identity, provenance and linkage of the samples.
4 The National Sample Archive (Appendix 4.3 and Figure 4.2)
The National Sample Archive in Scotland was established in 1998 and was the first standardised approach to archiving donor samples within SNBTS with all samples collected throughout Scotland being archived under one system. It is managed under Good Manufacturing Practice ( GMP) conditions and is prepared and stored in a controlled manner with good electronic record retention, availability and access. Although it cannot be considered equivalent to the testing of 'fresh' donor samples it does provide a high degree of confidence in sample integrity and provenance, and therefore also in the accuracy of the results obtained, when performing retrospective testing investigations.
5 Testing Methodology (Appendix 4.4)
In 1991 a serological test for anti- HCV was introduced for all blood donations. Currently all samples are still tested for anti- HCV but are also tested for HCV Nucleic Acid ( NAT).
Due to the volume of archive sample available it is not possible to perform both tests in this exercise. From a practical standpoint it is believed that the serological anti- HCV test is the more practical to complete. However it should be borne in mind that samples can and do degrade with time even under optimal storage and that continuous temperature monitoring records are unlikely to be available over a period of more than 30 years. Current generation virology assays are optimised for fresh samples and contemporary automated testing platforms whereas this testing will almost certainly have to be carried out using manual methods.
It is routine practice that any initial screen reactive (potential positive) test result is repeated in duplicate generating 3 results in total. This is because some donor samples can give rise to a false positive result in virological testing due to technical reasons. If 2 or 3 of the 3 tests are reactive then the sample is considered repeat reactive and the blood components are not issued.
With the highly sensitive screening assays used in SNBTS, >90% of repeat reactives will not be confirmed as 'true' positive infections. All repeat reactive donations are therefore referred for further testing and complex confirmatory algorithms are followed to ensure that only donors will real infections are informed of their infective status.
Due to the lack of sample volume it will not be possible to follow the above procedure for all samples and SNBTS would require to identify, trace and re-test all the donors of repeat reactive samples in order to confirm the veracity of the result (other than those who continued as donors after September 1991 who would have been tested for HCV and those already involved in lookback carried out for any HCV positive patients).
It needs to be borne in mind that whilst donors are, by definition, healthy individuals, there is still a high level of attrition over a 25-32 year period. Many of these people will have moved and some will be deceased and there will be no way of knowing this before we attempt to trace them. The emotional impact of contact on some donors and their families (particularly if the donor is deceased) needs to be taken into account.
6. SNBTS Donor and Donation Records (Appendix 4.5)
As noted above SNBTS introduced its first National IT system (Progesa) to manage donor, donation, testing and manufacturing and issue records electronically in 1998. Other IT systems had become available from the late 1980s/early 1990s but different systems were in use in different regions ( e.g. Dobbin/Lablan) and they were not as comprehensive as Progesa. Records prior to this were mainly paper based which in many cases were subsequently micro-fiched to reduce the bulk of paper storage.
For much of the 1984-1991 sample archive, there were no electronic records linking of testing to donors, donations collected, components manufactured and subsequently issued to hospitals.
It is unknown if access to paper or micro-fiche records remains possible for 1984-1991 but even if they are extant reconciling them is likely to be challenging and incomplete.
From the available information related to records it is likely that in a significant number of cases it would not be possible to unequivocally link a tested archive sample to a donor, to the components manufactured, or to the hospital to which they were issued.
7. Hospital Records (Appendix 4.6)
In the past hospitals were not required to maintain hospital blood bank records for the same length of time as SNBTS (this changed when the Blood Safety and Quality Regulations came into effect in 2005). Therefore if a confirmed anti- HCV positive result was obtained on testing of a 1984-1991 archive sample, even if traceable to the hospital blood bank, it is unlikely that in the majority of cases it could be traced to a specific patient. Even if or where the hospital blood bank records were extant, one would then require the clinical hospital records to confirm transfusion, identify the attending Consultant or General Practitioner and contact details for the patient. Most clinical records are not retained for this length of time (25-32 years), most of the attending clinicians will have retired and many of the patients will have moved or be deceased (50 % of blood components are administered to patients who die within 5 years of their underlying condition or age). It may well not be possible to identify whether a patient is alive or deceased prior to trying to contact them. It is very difficult to estimate the workload involved in trying to trace these records and individuals but it is likely to be a very labour intensive.
8. Logistics and Costs of Testing the 1984 - 1991 Sample Archive (Appendix 4.7)
For the purposes of this review, one scenario was explored to allow the logistics and indicative costs of testing to be estimated. Other scenarios are possible where additional resource could be employed to reduce the overall timescales.
From the foregoing it can be seen that testing the 1984-1991 would require considerable effort and resource in terms of identifying, retrieving and manually testing the samples, and reconciling the test results with the original donations and donors on the one hand and the components manufactured and issued to hospital blood banks on the other. As a reference point, SNBTS currently tests around 250,000 samples per annum using automated systems This work would require the testing of 2 to 2.5 million samples (10x the current annual SNBTS throughput) using manual methods for retrieval, sampling, testing and record reconciliation.
A separate team would need to be employed and trained to undertake this and currently we do not have additional laboratory facilities in which it could be undertaken. It is possible that with the opening of the new National Centre in mid-2017, one of the existing laboratories could be retained rather than decommissioned to provide space, but this would need to be formally explored. It would also be necessary to purchase additional equipment, validate the assays and protocols and recruit and train a dedicated team of suitably qualified staff before testing could be commenced.
The estimate in this scenario is that testing would take approximately a year to establish and 5-6 years to complete at a cost of between £8.2 million and £9.7 million. It should be borne in mind that this is only a high-level estimate of likely SNBTS costs only, the costs to other parts of NHS Scotland of searching hospital blood bank and patient records and tracing individuals is difficult to estimate.
9. Summary
The 1984-1991 sample archive was collected and stored between 25 and 32 years ago when assays, technology, record keeping, quality and IT systems were rudimentary and dispersed across 5 centres.
This review has identified that there are a number of challenges and limitations in testing the 1984-1991 archive:
- There are many concerns with the reliability of the archive in relation to sample identification, provenance and security of linkage to other key data.
- For many samples there will be insufficient sample volume to carry out testing to resolution.
- Linking tested samples to specific donors, components and the hospitals they were provided to will be difficult due to the variety of paper-based and early IT systems used at the time.
- An effort would need to be made to trace repeat reactive donors to inform them and carry out confirmatory testing. Most of these people will no longer be blood donors and may have moved or themselves be deceased.
- Hospital record retention does not extend back to the 1984-1991 period and it is likely to prove difficult to identify and trace which patients received specific components.
- A break in the chain of traceability in any of the above areas would negate the benefit of testing. In reality there are significant weaknesses in all areas. The likelihood of being able to trace significant numbers of infected patients through this route is small.
- This would be a major exercise, incurring costs of between £8m-£10m and requiring 5-6 years to complete.
Appendix 4.1a: Simplified Process Map of Blood Collection
Appendix 4.1b: Simplified Process Map of Donation Processing
Appendix 4.1c: Simplified Process Map of Donation Testing
Appendix 4.1d: Simplified Process Map of Order, Issue and Distribution
Appendix 4.1e: Simplified Process Map of Hospital Blood Bank Testing and Issue
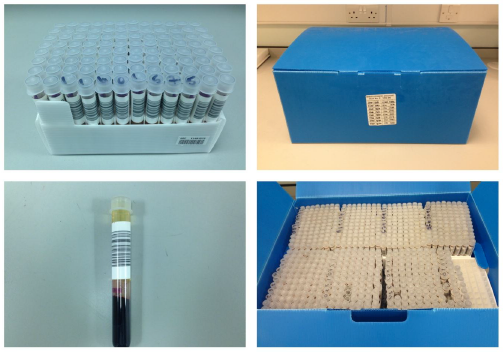
Appendix 4.2: Testing Sample Archive for HCV - 1984 - 1991 (see Figure 4.1).
All samples were collected, prepared and stored in the then five regional centres in Aberdeen, Dundee, Edinburgh, Glasgow and Inverness.
Archive samples were prepared by different means i.e. non-standardised in each region.
Archive samples were stored in a variety of containers - poly-tubes, 'small plugs', 96 well micro-plates, 96 deep-well micro-tubes, plugs, etc.
Record keeping and IT systems were not as developed as the systems in use today. Paper and/or electronic records are therefore poor and patchy. As with the storage containers a variety of record formats were used (for those that are available).
Archive sample volume varies, from a few micro-litres to about 1ml.
Most archive samples do not have bar-code identification labels or check digits with some having hand written donation numbers. The year of donation is not on the label and the numbers used were repeated each year, for example number range started at 000001 in 1984 would be repeated as 000001 at some time in the future. Those on micro-titre plates will not be individually numbered - would depend on whether plate maps or copies of test results are available (we know many are missing) for individual sample identification.
Many of the archive samples were prepared manually and this is known to be subject to human error (in sampling and transcription).
Archive sample condition (suitability) for testing is unknown, i.e. we don't know how well they have been stored (thawed and/or re-frozen) or how many temperature excursions there have been.
Current assay package inserts require frozen thawed samples to be well mixed and centrifuged before testing - some package inserts require ultra centrifugation. This is clearly not possible with micro-titre plates or for large scale screening which this exercise would require. However such testing is acceptable for small numbers such as the occasional investigation.
Success (based on experience) in trying to locate samples from this era is relatively poor.
We cannot test the archive samples on automated platforms, they will have to be tested manually.
Many of the poly-tube archive samples were removed from storage trays and placed in plastic bags. They would take considerable time and effort to sort.
The entire Glasgow, Aberdeen, Inverness and Dundee sample archives were sent to Edinburgh around about 1998 - many records are missing.
The staff who dealt with this at the time have long since left the organisation. No 'organisational memory' available to assist with process.
Not possible to create/retrieve lists of all donations collected by Date of Withdrawal ( DOW) - no way to reconcile what we have in archive against what was actually collected and tested at the time.
Of 19 requests for 'look-back' samples covering this time period only 11 could be found.
Some archive samples, directly relevant to this exercise, may have already been removed from the archive for previous look-back investigations.
We may know what archive samples have been stored - but we don't know what samples are missing. It may not be possible to identify them so the exercise cannot ever be entirely complete.
Figure 4.1: Photograph of the 1984 - 1991 Sample Archive.

Figure 4.2: Photograph of the post-1998 National Sample Archive.
Appendix 4.3: Testing Sample Archive for HCV - post-1998
First established 1998.
PPT gel tube stores plasma and red cells separated by a gel interface after centrifugation - so no cellular contamination.
Good volume for further testing - initial sample 6-7ml on collection and prior to routine NAT testing.
Bar coded sample (and eye-readable) with appropriate check digits incorporated - provides confidence when sampled and tested on automated instruments.
Low risk of contamination from other samples as disposable tips used.
It is used as routine NAT testing sample prior to be used for archive purposes.
All archive samples prepared in Edinburgh P&T laboratory - standardised approach with no variation from different sites.
Good electronic records - can confidently link archive sample to test results through to full donor records.
For each sample we know position in archive rack, in which box rack stored, on which pallet box stored, and location in commercial store. Fairly easy to locate if required although we need permission to access commercial store as they have to retrieve pallet using forklift truck.
Prepared and stored to contemporary GMP standards.
Can be tested on automated assay analysers if required (not sure this is actually true given need to remove debris).
Current process provides confidence that the sample is from identified donor.
Samples have been stored well throughout their archive life (controlled).
We have a good success rate in finding and retrieving these samples when required to do so.
Stored in commercial cold store under contract to an agreed specification.
Very happy to use these samples and have high confidence in them (for testing small numbers)
Requirement of current package inserts to have frozen/thawed samples ultra centrifuged to remove any debris prohibits large scale screening on a routine basis. This is a complication that needs to be explored further as large scale screening of current archive may not be as easy as initially thought.
Appendix 4.4: Testing methodology
There are two broad methods of testing samples for HCV, serology (antibody response) and nucleic acid testing ( NAT).
NAT is more sensitive and would pick detect donors with active infection who are more likely to have transmitted infection, however with the age and unknown storage conditions of samples the titre may have dropped off from the original titre leading to at least some loss of sensitivity (increased false negatives). Practical issues associated with NAT however include the need to make artificial pools which would have to be done manually given the variety of sample storage containers, with manual transcription of sample numbers introducing another level of possible error. Also there would be a sample volume issues for NAT (we routinely test 0.5ml) and any positive pools would require further resolution/further samples which are unlikely to be available.
Serological assays require less sample volume. The sensitivity and specificity of current HCV antibody screening assays are assay dependent but are in the region of 99.7 - 99.9% . That of course is based on testing samples as defined in the kit insert (usually 7-14 days old and stored at 4 degrees C). We're not sure we can say what impact the storage of samples over 25-30 years would have on the sensitivity and specificity of the test though in broad general terms these are likely to drop somewhat with old samples.
So in light of the age and variable storage of the samples,
either testing approach will lead to a higher level of false
negatives (reduced sensitivity) and/or false positives (reduced
specificity) compared to fresh samples.
An initial screen reactive result is not a confirmed result
and a donor couldn't be classified as
HCV positive based on
this.
Repeat tests are normally performed in duplicate, considered repeat reactive if 2 of the 3 tests are reactive. All assays have samples that will be non-specific false positives - so being reactive is not confirmed as due to HCV.
Where there is sufficient volume (unlikely) repeat reactives would be referred for confirmatory testing. Negative, indeterminate or confirmed possible outcomes.
Unable to test actual donation to ensure correct donor has been identified.
Assume we will begin to trace donor based on these results. Need to explore Dobbin/Lablan records to identify donor.
Uncertain whether donor will be at same address and some will themselves be deceased.
Significant workload for medical staff in following up donors (this has not been quantified and will depend on test results).
Appendix 4.5: Records - SNBTS
During the period 1984 to 1991 IT systems were rudimentary or non-existent, both within Donor Services, Testing, Components and Distribution. Paper records were primarily maintained and due to their bulk and the space required to store them many were micro-fiched and are therefore difficult to access now (if they can be located at all and not electronically).
Electronic Donor database system did not become available until the late 1980s and these were more rudimentary than the systems currently in use today. Dobbin was the system in use throughout SNBTS with the exception of Glasgow where a system called LabLan was utilised.
When Progesa was introduced in 1998, donor records were migrated from both LabLan and Dobbin. However due to the number of records to be migrated it was agreed that the details of only the most recent donation for every donor would be transferred into Progesa. Therefore many, if not most, of the donation details for the period 1984 - 1991 will not be available electronically now in eProgesa.
Currently, when investigating possible transfusion transmitted infections or adverse reactions, the IT department write an extraction programme to interrogate Dobbin records for each and every request (with limited success).
Many of the paper records are stored off-site in commercial stores and access to them and locating what is needed would be logistically challenging.
Paper records of components manufactured were maintained, we don't know if these are still available or traceable.
Issues of components to hospitals were simply recorded manually in ledgers, again we don't know if these are still available.
In summary, records for this period are poor, patchy and in some cases no longer extant.
Appendix 4.6: Records - Hospitals
The timescales Hospitals are required to retain patient records are detailed below.
Adults
6 years from date of last entry.
3 years from date of death.
Children
From birth until 25 th birthday.
If date of entry is at aged 17 years until 26 th birthday.
3 years from death.
Any archive sample from 1984-1991 SNBTS tested now and found to be positive is therefore, on balance, unlikely to be able to be traced to a specific patient because it is likely that hospital blood banks records and patient notes from the time have been destroyed.
Many of the patients may have died for other reasons or the illness they were admitted for.
Many patients may have moved address.
The current universal NHS Scotland CHI identification system was not in place during this time period.
Many changes to hospitals have occurred since this time and it is likely that this would also impact on record availability and traceability (closure, consolidation and rationalisation of services for example).
Appendix 4.7: Logistics and Costs of Testing
This is based on manually testing up to 1500 samples per day in a manual system.
Bring 3 pallets/cages of frozen samples from commercial store into SNBTS hands per day.
Need freezers to store within SNBTS.
Estimate 6 staff required to remove sufficient samples from pallet prior to each day testing.
Samples will require to be thawed and centrifuged - not possible for micro-titre plates unless decanted into other tube - this would be extremely onerous and alone could take 6 staff per day.
Need to prepare lists of samples available for testing - this would require to be done manually i.e. hand written - this would be a lengthy process and prone to transcription error.
Set up tests manually - need to identify and procure a suitable assay - this would need to be validated for the method and samples used. It is difficult to say whether any manufacturer would support their test being used for this purpose. Assume need 6 staff to set up and test around 1500 samples per day by hand.
Need equipment to perform manual test - no positive sample ID available - wash, add reagents and read results. Printout of results would not have donation numbers. Unable to link results to donation number electronically. We don't have this type of equipment for large scale screening - would need to procure and validate.
Don't have space to locate and operate this equipment - a specific laboratory would need to be created for this purpose. Need cold room space for thawed samples and reagents. Could be considered for 'development' space within new National Centre (not available until middle of 2017).
Manual reconciliation of results would be onerous, labour intensive and time consuming.
Recruitment and training of staff must also be considered and set up time is likely to take a year.
Test is likely to cost around £2 and test around 400,000 samples per year - so around £800,000 per year.
Around 20 new staff would need to be recruited and trained. Say 16 at Band 6, 3 at band 7 and 1 at band 8 from 6 years (inclusive of set up year) = £700,000 per year.
Approximately 2-2.5 million samples - so a year set up and 5-6 years to complete testing = £8.2 million - £9.7 million.
Please note there are likely to be other costs not covered in the above analysis - for example the full costs of laboratory set up and equipment purchase and the costs to the broader NHS Scotland of trying to trace patients through hospital blood bank and patient records.
By increasing staffing numbers, equipment and laboratory space it would be possible to reduce the overall timescales with some increase in costs.
Contact
Email: Robert Girvan, robert.girvan@gov.scot
There is a problem
Thanks for your feedback