Report of the National Cremation Investigation by Dame Elish Angiolini DBE QC
Investigation findings and recommendations following an investigation across crematoria in Scotland who did not routinely return ashes to families following the cremation of infants.
18 Annex: Report by Dr Julie Roberts, Forensic Anthropologist
Anthropology Report
Report of Julie Ann ROBERTS BA (Hons), MSc, PhD
Occupation Forensic Anthropologist and Archaeologist
at
Cellmark Forensic Services
Unit B1, Buckshaw Link, Ordnance Rd, Chorley, Lancashire, PR7 7EL
Dated the 31st day of May 2016
Signature…………………………………………………….
Laboratory Reference Number: CFS/935214/16
Customer Reference: National Cremation Investigation
Contents of Report
1. Qualifications and Experience
2. Background
3. Scope and Purpose
4. Technical Notes
5. Skeletal Development in the Foetus, Neonate and Infant
6. The Cremation of Human Remains
7. The Survival of Foetal, Neonatal and Infant Remains During and After Cremation
8. The Relationship between Methods of Cremation, Survivability of Remains and Recovery of Ashes
9. Distinguishing between Adult, Neonatal and Infant Remains that were Cremated Together
10. Conclusions
1. Qualifications and Experience
I have been employed as Scientific Lead and Team Leader for the Anthropology, Archaeology and Ecology Department at Cellmark Forensic Services since September 2010. I hold a Doctoral degree (PhD) in the subject of Forensic Anthropology from the University of Glasgow, a Master of Science degree (MSc) in Osteology, Palaeopathology and Funerary Archaeology from the University of Sheffield, and a Bachelor of Arts degree (BA Hons) in Archaeology and Ancient History from the University of Manchester.
I have worked as a Biological Anthropologist and Archaeologist for approximately 21 years and I have approximately 17 years forensic casework experience, specialising in the excavation and examination of decomposed, burnt, fragmented and commingled human remains from scenes of crime. I specialise in the analysis of fragmented, burnt and commingled bone and I have undertaken research on the taphonomy of burnt human bone, and the colour changes and fracture patterns which occur as bone is burnt. I researched and examined Bronze-Age cremation burials at Glasgow University for approximately eight years and have published extensively on this subject in archaeological journals. I have undertaken a wide range of forensic casework relating to burnt human remains which includes the recovery, examination and reconstruction of burnt, fragmented and commingled remains from fatal fires in houses, cars, aircrafts and military vehicles. I have also recovered, examined and reconstructed burnt fragmented bone from victims of war crimes, terrorist incidents, individual and multiple homicides in the UK where bodies have been burnt in attempts to dispose of evidence. I have examined burnt adult and juvenile bone from archaeological and forensic contexts and have also been required to distinguish between burnt human and non human bone from fatal fires and large bonfires. I have produced numerous witness statements relating to burnt and fragmented remains and given evidence in court and at inquest on my anthropological examinations of burnt remains.
I am professionally accredited by the Royal Anthropological Institute as a Forensic Anthropologist Cert FA-I (most senior level) and I am registered with the National Crime Agency as an Expert Advisor in Forensic Anthropology and Archaeology. I am a member of the Forensic Expert Group advising UK DVI on Forensic Anthropology, a Fellow of the Royal Anthropological Institute, a member of the International Academy of Legal Medicine and the Forensic Anthropology Society of Europe, and a member of the steering committee for the British Association for Forensic Anthropology.
2. Background
On the 22nd January 2013, The Right Honourable Dame Elish Angiolini DBE QC was appointed to undertake an independent inquiry into practices surrounding the cremation of pre- term and new born infants at Mortonhall Crematorium in Edinburgh. As part of the Mortonhall Investigation I was asked to provide expert opinion on the skeletal development of the foetus and neonate, how the cremation process affects the body, the survivability of foetal remains during and after cremation; the relationship between the survival and recovery of remains and the methods used to cremate and retrieve them from the cremator; and the accuracy of advice provided by Funeral Directors and crematoria staff at that time. I produced my findings in two statements, dated 7th January 2014 and 10th March 2014, which appeared in full in Annex C and Annex D (respectively) of the Mortonhall Investigation Report, produced by Dame Elish Angiolini on the 14th April 2014. The report by Dame Elish was subsequently published by City of Edinburgh Council on the 30th April 2014 (The City of Edinburgh Council, 2014).
Whilst the independent investigation relating to Mortonhall was progressing, the Scottish Government established The Infant Cremation Commission, chaired by Lord Bonomy. The Commission was tasked with reviewing " current policies, guidance, practice and legislation in Scotland in relation to the handling of all recoverable remains (ashes) following the cremation of babies and infants and to make recommendations for improvement and change" (The Scottish Government, 2014). Lord Bonomy's Infant Cremation Commission Report, published by the Scottish Government on the 17th June, 2014, provided national recommendations for future improvements ( ibid).
On the 17th June 2014, as a result of the findings from the two investigations, the Minister for Public Health announced the establishment of a National Investigation into infant cremations in Scotland as it was acknowledged that families from some areas of Scotland were still seeking answers (The Scottish Government, 2015). The aim of the National Investigation was therefore to "provide every parent whose baby was cremated in Scotland with the same opportunity to have their concerns regarding their cases investigated and to get an individualised response" ( ibid). Dame Elish Angiolini was appointed to lead the investigation and, as part of the investigation she requested that I meet with her and Claire Soper from the National Cremation Investigation Team, to discuss the provision of a forensic anthropology expert report.
During my subsequent discussions with Dame Elish and Claire Soper it became apparent that in certain locations concerns remained regarding the cremation of pre-term babies and infants. In one example at Aberdeen crematorium there was a policy of "no ashes of children aged 2 years and younger" being returned to parents. At the same crematorium it was reported that non-viable fetuses, neonates and infants were being cremated alongside adults, with more than one coffin at a time being placed in the cremator. In these cases it was apparently not possible to identify who the adults concerned had been.
3. Scope and Purpose of Report
In January 2016, I met with Dame Elish and Claire Soper. During the meeting it was agreed that I would assist the National Cremation Investigation by providing expert opinion on the following:
- The skeletal development of infants from age 40 weeks to 24 months.
- The size and appearance of burnt skeletal remains from infants of specified ages within that range.
- The amount of ash that would be produced from the cremation of infants of specified ages within that range.
- Whether it would be possible to distinguish between adult skeletal remains and infant skeletal remains if they had been cremated together.
- The possibility that the skeletal remains of infants up to two years of age might not survive the cremation process.
It was also confirmed that the following specific data should be included in my report:
- A summary of skeletal development in the neonate and infant between full term and two years
- A range of expected sizes of bones at age 40 weeks gestation, 3 months, 6 months, 12 months, 18 months and 24 months.
- Forensic drawings of cremated foetus and neonate bones reproduced from photographs taken at Seafield and Warriston Crematoria in Edinburgh and Parkgrove Crematorium in Douglasmuir. Full details of which images were used and how they were reproduced can be found in Sections 4.2 and 7.2.
My report also contains information on the cremation process and how it affects the body, the survivability of neonatal and infant remains during and after cremation, and the relationship between the survival and recovery of remains and the methods used to cremate and retrieve them from the cremator. Finally, consideration is given in the report as to whether there is any scientific credibility in the notion that there would be no remains or ashes [1] left following the cremation of infants aged up to two years. This was thoroughly researched as that belief (or official position) may have informed the policy decisions made at certain crematoria with regard to the return of ashes and information given to bereaved parents.
4. Technical Note
This report has been produced as a "stand alone" document for the National Cremation Investigation. It does however contain sections of text derived from the two reports I produced previously for the Mortonhall Investigation (The City of Edinburgh Council, 2014). These sections relate primarily to the cremation process and the effects it has on the body, the growth and development of the foetus in-utero, the physical appearance and dimensions of cremated foetal and neonatal bones and the ability of them to survive the cremation process. This has been supplemented with additional data on neonates and new data on infants aged up to two years at death.
4.1 Appendices and Archive
Diagrams of the skeletons of a neonate and a young child are provided for reference purposes in Appendix One. A glossary of terminology used in the report is provided in Appendix Two, a list of sources of metric data for foetal and infant remains is provided in Appendix Three, and a bibliography of texts referred to in this report can be found in Appendix Four. A full record of the work undertaken within the laboratory in relation to this work has been retained in the archive at Cellmark Forensic Services, Chorley, and this can be made available on request providing sufficient notice is given.
4.2 Reproduction of Photographic Images as Forensic Drawings
It was agreed that it would be of value to include a graphic representation of the some of the surviving neonatal and foetal remains identified at the private crematoria, Warriston, Seafield and Parkgrove. Due to the sensitive nature of the subject matter, however, Dame Elish and her team did not wish to show the original photographs taken at the crematoria. After discussion it was agreed that the best way to show the images would be for a suitably qualified and accredited expert to produce hand-drawn or electronically generated "drawn" images of the bones that could be seen in the selected photographs.
This work was undertaken by the Multimedia & Evidential Imagery Team, Specialist Operations Regiment, Royal Military Police, using Adobe Photoshop CS5 and a Wacom Tablet.
4.3 Terminology relating to Foetal Age and Infancy
It should be noted that there is a difference between gestational and conceptional or foetal age. Gestational age refers to the length of pregnancy after the first day of the last menstrual period ( LMP) and is usually expressed in weeks and days. Conceptional age is the true fetal age and refers to the length of pregnancy from the time of conception (Mongelli, 2012). Fertilisation can not occur till ovulation has occurred approximately 14 days after the first day of the menstrual period. As such conceptional age is always approximately two weeks behind gestational age ( ibid). Gestational age is more frequently used because the actual day of conception is often unknown, whereas the LMP can usually be determined.
Infancy, as defined by Bogin (1999) refers to the three years of human life following birth, characterised by dependency prior to the child adopting a "close to adult diet" and "some independence of social interaction" (Black and Maat, 2010: 82).
For further terminology relating to foetal and infant age see Appendix Two.
4.4 Comparative Data and Related Research
A great deal of the literature relating to cremated bone is based on experimental research using archaeological human remains or modern animal remains. This information covers a range of topics and is easily accessible. However, primarily for ethical reasons, there is has been little research involving modern cremated human adult remains and even less focusing on foetal neonatal and infant remains. Because of this there is a limited amount of scientific data available for reference purposes when it comes to providing an evidence based opinion on the survival of these remains during and after cremation. When considering this, the limited reference data must therefore be supplemented by knowledge of skeletal development, how cremation affects the body, visual examination of relevant images from modern crematoria and familiarity with findings from research on non-human and ancient human remains.
5. Skeletal Development in the Foetus, Neonate and Infant
Detailed information relating to the development of the foetus can be found in my previous two reports dated 7th January and 10th March, 2014, produced for the Mortonhall Investigation (The City of Edinburgh Council 2014). This report includes an abridged summary of foetal development and provides additional detail on the skeletal development of the neonate and the infant from age 40 weeks through to 24 months.
5.1 Development and Ossification of the Foetus in-utero
Bone develops from the primitive mesenchymal tissue of the embryo in a process called ossification. There are two types of ossification; intramembranous and endochondral (Scheuer and Black, 2000: 21-24). The essential difference between the two is the presence or absence of a cartilaginous phase. The majority of bones, including the limb bones, vertebrae, ribs and basi-cranium are formed by endochondral ossification (Scheur and Black, 2000: 24; White and Folkens, 2005: 46; Sanders, 2009). In endochondral ossification a cartilage template composed mainly of collagen is first formed out of the tissue membrane, this then ossifies (turns to bone) due to the action of osteoblasts and osteoclasts.
The bone will simultaneously increase in diameter and length, as compact and cancellous bone is formed [2] . Compact bone is composed of parallel columns along the long axis of the bone and it forms the shaft or cortex (outer surface). Cancellous bone is arranged in a lattice structure orientated along the lines of stress and it provides structural strength within the bone. Lengthways growth in the long bone is achieved by means of a growth plate at the end of the shaft of the bone. New bone is deposited between the growth plate, also known as the epiphyseal plate, and the end of the diaphysis (the shaft) which is termed the metaphysis. (White and Folkens, 2005: 46).
Scheuer and Black cite the clavicle as being probably the first bone in the human body to show evidence of bone development in the sixth week of foetal life (2000: 23). In a study of ossification of the limb bones in 728 foetuses ranging in age from 8 to 26 weeks, Bagnall et al. found that primary ossification centres showed at approximately 9 weeks of conceptional age (Bagnall et al., 1982). They also observed that there was a predictable order to this ossification whereby the centre of the humerus appeared first followed by the femur, radius and ulna which appeared simultaneously, the tibia and then lastly the fibula ( ibid).
On a molecular level, bone tissue is a composite of organic and inorganic material, protein and mineral. The protein is collagen which constitutes about 90% of the bones organic content. The mineral component is hydroxyapatite, a form of calcium phosphate. Crystals of this mineral impregnate the collagen matrix to form a weave of protein and minerals. The mineral component gives the bone its hardness and rigidity, whilst the protein component is rubber-like and flexible (White and Folkens, 1991: 19). The composition of bone is highly relevant when considering the effects of cremation on bone and expectations regarding its survival of the process.
The rate of growth differs between the upper and lower limb bones. In early development the upper limb bones are longer than the lower limb bones due to their earlier ossification and faster growth rates (Sanders, 2009). From 19 weeks gestation until birth, however the lower limb bones grow faster than the upper limb bones and the reverse becomes true (Watkins and German, 1992, in Sanders, 2009). Studies have shown that there are also differences between the growth rates of bones on the right and left sides of the body in utero, with growth of the humerus, tibia and fibula being favoured on the left side and growth of the femur being favoured on the right (Bagnall et al., 1982). In terms of recognition of skeletal elements, Scheuer and Black (2000) note that by 12-13 weeks gestation bones such as the femur are distinct enough for identification (Sanders, 2009). The weight of the foetal skeleton will increase with age, with the greatest proportionate contribution being the skull (Trotter and Hixon, 1974).
5.2 Skeletal Development in the Infant: From Birth (40 weeks Gestation) to 24 months Immediately after birth, post-natal ossification centres appear which continue to develop into early adulthood. These centres fall into two groups; the primary centres of the bones of the wrist and ankle, and the secondary ossification centres of the ribs, vertebral column, sternum, shoulder and pelvic girdles and the long bones of the limbs ( ibid, 2000: 8). Together with the teeth, the neurological system is the quickest to develop and skeletally this is reflected in the early maturation of the cranium and the vertebral column (Black and Maat, 2010:82).
The primary centres of the bones of the wrist appear from two to four months after birth and (although they are very small) by 2 years of age, three of them, the capitate, hamate and triquetral are present and identifiable ( ibid: 328). In the ankle, two of the tarsal bones, the calcaneus and the talus will be present at birth and by the age of 2 years primary centres for the cuboid, the lateral and possibly the medial cuneiform will be present ( ibid: 449).
Some of the bones in the skull which are separate at birth will join at around 40 weeks gestation, for example the tympanic ring (a small ring of bone) will fuse onto the outside of the temporal bone around the ear. Some of the cranial sutures will also start to fuse in infancy, for example in the frontal bones, and the anterior fontanelle will be closed by age two years in the majority of cases (Scheuer and Black, 2000: 107).
The secondary sites of ossification appear after birth, developing within the cartilaginous epiphyses which are separated from the metaphysis by the growth plate. Controlled by hormones and genes and influenced by other factors such as health and nutritional status, bone growth continues at the metaphysis until such a time that it has reached its predetermined size. Cells at the growth plate then stop dividing and the primary and secondary sites of ossification (the main part of the bone and the epiphysis) fuse together in a process called epiphyseal fusion or closure (Biswas and Iqbal, 1998: 59; White and Folkens, 2005: 47). Once the epiphyses have closed the development of that bone is complete. This occurs from early infancy through to the age of up to 29 years when the medial end of the clavicle finishes development (Scheuer and Black, 2000).
Infancy is the period in which the child will show the most rapid growth of any of the postnatal stages (Black and Maat, 2010). According to Black and Maat, the child will increase its length by around 50% and double its birth weight in the first year after birth ( ibid: 82). Doyle (2016) summarises "Normal term neonates generally lose 5 to 8% of birth weight in the days after delivery but regain their birth weight within 2 weeks. They then gain 14 to 28 g a day until 3 months, then 4000g between 3 and 12 months, doubling their birth weight by 5 months, tripling it by 12 months and almost quadrupling it by 2 years (see also Table Eight, this report).
Tables One to Six, present the maximum lengths of six bones which are easily identifiable in the neonate and infant. These are mean measurements of unburnt bone. In the long bones they relate to the diaphysis alone, i.e. the shaft without the epiphyses. Where measurements are presented between the male and female columns this indicates that the sex relating to the value was not stipulated. Information on the individual reference sources is given in Appendix Three.
Table One: The Frontal Bone of the Cranium (Length: frontal chord)
| Age |
Maximum Length (mm) Reference |
|
|---|---|---|
| Male |
Female |
|
| 40 weeks |
54.8 |
Fazekas and Kosa, 1978 |
| 3 months |
81.9 |
Young, 1957 |
| 6 months |
89.3 |
Young, 1957 |
| 12 months |
99.6 |
Young, 1957 |
| 18 months |
- |
- |
| 24 months |
109.2 |
Young, 1957 |
Table Two: The Femur
| Age |
Maximum Length (mm) Reference |
|||
|---|---|---|---|---|
| Male |
Female |
|||
| 40 weeks |
75.40 |
74.4 |
70.7 |
Fazekas and Kosa, 1978 Trotter and Peterson, 1969 |
| 3 months |
100.7 |
100.8 |
Maresh, 1970 |
|
| 6 months |
112.2 |
111.1 |
Maresh, 1970 |
|
| 12 months |
136.6 |
134.6 |
Maresh, 1970 |
|
| 18 months |
155.4 |
153.9 |
Maresh, 1970 |
|
| 24 months |
172.4 |
170.8 |
Maresh, 1970 |
|
Table Three: The Humerus
| Age |
Maximum Length (mm) Reference |
|||
|---|---|---|---|---|
| Male |
Female |
|||
| 40 weeks |
65.2 |
64.9 |
61.2 |
Fazekas and Kosa, 1978 Trotter and Peterson, 1969 |
| 3 months |
80.6 |
80.2 |
Maresh, 1970 |
|
| 6 months |
88.4 |
86.8 |
Maresh., 1970 |
|
| 12 months |
105.5 |
103.6 |
Maresh, 1970 |
|
| 18 months |
118.8 |
117 |
Maresh, 1970 |
|
| 24 months |
130 |
127.7 |
Maresh, 1970 |
|
Table Four: The Tibia
| Age |
Maximum Length (mm) Reference |
|||
|---|---|---|---|---|
| Male |
Female |
|||
| 40 weeks |
66.8 |
65.2 |
60.8 |
Fazekas and Kosa, 1978 Trotter and Peterson, 1969 |
| 3 months |
81.9 |
80.8 |
Maresh, 1970 |
|
| 84.83 |
84.95 |
Gindhart, 1973 |
||
| 6 months |
91 |
88.9 |
Maresh., 1970 |
|
| 99.26 |
97.06 |
Gindhart, 1973 |
||
| 12 months |
110.3 |
108.5 |
Maresh, 1970 |
|
| 119.57 |
117.08 |
Gindhart, 1973 |
||
| 18 months |
126.1 |
124 |
Maresh, 1970 |
|
| 135.53 |
134.24 |
Gindhart, 1973 |
||
| 24 months |
140.1 |
138.2 |
Maresh, 1970 |
|
| 150.14 |
148.08 |
Gindhart, 1973 |
||
NB the Gindhart reference data was obtained from radiographs whereas the Maresh standards are measurements of dry bone in infants who had died. The consistently shorter length of the bones in the Maresh study may be a reflection of the poor health of the children who died, although this cannot be confirmed and other population specific factors may be involved.
Table Five: The Clavicle
| Age |
Maximum Length (mm) Reference |
|
|---|---|---|
| Male | Female | |
| 40 weeks |
44.1 |
Fazekas and Kosa, 1978 Yakoni et al., 1985 |
| 3 months |
44.4 |
Black and Scheuer, 1996 |
| 6 months |
54.1 |
Black and Scheuer, 1996 |
| 12 months |
59.5 |
Black and Scheuer, 1996 |
| 18 months |
63 |
Black and Scheuer, 1996 |
| 24 months |
66.5 |
Black and Scheuer, 1996 |
NB the Yakoni et al. reference data was obtained from ultrasound scans
Table Six: The Scapula (length / height)
| Age |
Maximum Length (mm) Reference |
|
|---|---|---|
| Male | Female | |
| 40 weeks |
35.5 |
Fazekas and Kosa, 1978 |
| 34.8 |
Hrdlicka, 1942 |
|
| 46.5 |
Vallois, 1946 |
|
| 3 months |
- |
|
| 6 months |
49.2 |
Saunders, 1993 |
| 12 months |
60.4 |
Saunders, 1993 |
| 18 months |
- |
|
| 24 months |
67.8 |
Saunders, 1993 |
| 61 |
Vallois, 1946 |
|
5.3 Factors Affecting the Development and Maturation of the Skeleton in utero and during Infancy
Rates of increase in the size and maturity of bones differ between the sexes and this is evident before birth. There is also a difference in the timing of ossification of bones and mineralisation of teeth (Scheuer and Black, 2000:4). In their research Bagnall et al. (1982) observed that the female foetus is in advance of the male in terms of skeletal maturation after 21 weeks. After birth skeletal maturity continues to be more advanced in girls than boys but bone mineral density is significantly less in girls than boys, the latter having a higher mineral density and larger long bones ( ibid)
Sanders (2009) summarised a number of studies which focused on femoral lengths of neonates and foetuses of different ancestries. In one study (n=450), it was found that the femoral length of Indian neonates was significantly longer than that of Malaysian and Chinese neonates (Lim et al., 2000 in Sanders, 2009:18). In another study which took femoral measurements by ultrasound from 39 Asian, 31 Black, and 100 White foetuses of 15 to 20 weeks gestation, it was found that the femur lengths of the Asians were shorter than expected and those of Black foetuses were longer than expected. (Shipp, 2001 in Sanders, 2009: 18).
In a research project which examined the weight, density and percentage ash weight of bones from foetuses through to elderly adults, Trotter and Hixon (1974) found that the unburnt bones of Negroid foetuses were on average heavier than those of the Caucasoid foetuses and the bones of the males were generally heavier than those of the females. These differences were not statistically significant, but there were significant differences between the lengths of the Negroid and Caucasoid long bones, the former being longer than the latter in four types of long bone tested ( ibid).
Whilst genes play a major role in the development of a foetus in utero, other influences can affect growth and development greatly. Black and Maat (2010) list the following as factors that influence prenatal growth: Maternal weight, maternal age, health and nutritional status, blood pressure, intra-uterine constraints, parity, smoking, alcohol / drugs, emotional status, environmental pollution and altitude. Similarly, Lobo and Zhaurova (2008) stated that "It is difficult to overemphasize the importance of prenatal environment to a developing fetus". They were speaking primarily with reference to birth defects but in addition to these they also found that stillbirths and low birth weights were associated with smoking during pregnancy.
It should therefore be taken into account when looking at unburnt and burnt foetal and neonatal skeletal remains that the pregnancy may have ended in spontaneous abortion or stillbirth because the baby was not developing normally. As such the bones may be smaller and perhaps not as well developed as they would be in a healthy foetus of the same gestation.
Many of the same factors which affect foetal and neonatal growth play a major role in the growth and development of infants (de Onis et al, 2009; Uysal, 2006; Simpson and Kunos, 1998; Goodman et al., 1984). The effects of in utero influences can be carried over into infancy and even into adulthood ( ibid; Malina et al, 2004) and whilst maternal health and lifestyle choices may have less of a direct impact on the child once it is born, they are still a consideration (de Onis et al., 2009). Infant growth and development are influenced by other factors too, such as physiological or psychological stress which might include childhood illness, periods of malnourishment and an individual's resistance to stress (Roberts and Manchester 1997; Uysal, 2006; Ameen et al 2005). These factors are inextricably linked with the social, political, environmental and economic situation that the infant is born into (Behrents and Broadbent, 1984).
Studies by the World Health Organisation between 1997 and 2003 examined the growth and development of 882 infants from birth to 24 months (and beyond) from six diverse geographical regions; Brazil, Ghana, India, Norway, Oman and the USA. External factors that they examined included duration of breast feeding and smoking during and after pregnancy. They concluded that "healthy children from around the world who are raised in healthy environments and follow recommended feeding practices have strikingly similar patterns of growth" (de Onis et al, 2009). This suggests that, in their study at least, maternal and external factors outweighed genetic factors such as sex and ancestry.
5.4 Dental Development
Although teeth are not part of the skeleton it is important to mention their development in this context as they are capable of surviving the temperatures attained during the cremation process, particularly when they are un-erupted and protected by the jaw.
The onset of tooth formation starts with the first deciduous [3] incisor between 14 and 16 weeks after fertilization (16 and 18 weeks gestation). This is followed 2 weeks later by the second incisor and then a week after that the canine starts to form. Deciduous first molars are initiated around 15 weeks after fertilisation and deciduous second molars 3-4 weeks after that when the foetus is in its 18th -19th week of life (Hillson, 2002: 121).
The first permanent molar also starts forming in utero around 28-32 weeks after fertilization with the lower molars starting to develop slightly earlier than the upper. The other permanent teeth do not start to develop until after birth.
At birth the neonate will have a full set of un-erupted deciduous dentition comprising upper and lower central and lateral incisors, canines, and first and second molars (Ubelaker, 1989). All of the deciduous dentition usually erupts within the first two and a half years after birth and by this time the permanent incisors, canines and first molars will also be developing in the jaw (Hillson, 2002: 1139; Ubelaker 1989). An infant aged two, therefore, will have mixed dentition some of which is capable of surviving cremation (See Section 7).
As with skeletal development, girls are in advance of boys, with various studies finding a difference of as much as a year (Hillson, 2002: 125). The difference between the sexes is greater in black girls and boys (double that seen in whites) and black children achieved each stage of dental development on average 5% earlier than white children ( ibid). A study by Simpson and Kunos (1998), which included infants from birth to aged 24 months, also identified health as a factor in tooth development, particularly in the canine which proved to be more affected by health and hormonal status than the other teeth.
6. The Cremation of Human Remains
The sequence of cremation can be described as follows:
1. Ignition and burning of coffin and outer layers of body
2. Drying of the "wet" parts of the body followed by the burning of the contents of the thoracic, cranial and abdominal cavities
3. Completion of the burning of combustible parts
4. Calcination of bones
5. Cooling of ash remains and ash processing to produce a final ash of small particles
(Davies and Mates, 2005: 132)
The discussion below focuses in more detail on the changes which the human body undergoes when it is cremated. It is out-with my sphere of expertise to comment on the technical aspects of the cremation process, for example how the cremator works and various legislation surrounding the cremation process. Expert opinion on this is provided in the specialist statement of Dr Clive T Chamberlain produced for the Mortonhall Investigation (The City of Edinburgh Council 2014) and in a further publication The Encyclopaedia of Cremation (Davies and Mates, 2005).
6.1 "Ashes" versus "Cremated Remains"
The distinction between the terms "ashes" and "cremated remains" and the confusion that often surrounded the definitions was discussed previously in my report dated 7th January 2014 which I produced for the Mortonhall Investigation (The City of Edinburgh Council, 2014). Since the publication of that report and the work of the Infant Cremation Commission, this issue has largely been resolved.
In his report, the Rt. Hon Lord Bonomy recommended the following:
"2.3 The "ashes" which the Cremation Authority is obliged to give into the charge of the person who applied for the cremation if he so desires should be defined in legislation as "all that is left in the cremator at the end of the cremation process and following the removal of any metal". That should not preclude the applicant from consenting in advance to the removal of metals, such as coffin nails and artificial joints, and their separate disposal, including as part of a metal recycling scheme. (7.21) (The Scottish Government, 2014).
Subsequently, section 45 of the Burial and Cremation (Scotland) Act 2016, passed on 22 March 2016, stipulates:
"(1A) In this Act 'ashes' means the material (other than metal) to which human remains are reduced by cremation.
(1B) In this section "human remains" includes, where remains are clothed, in a coffin or with any other thing, the clothing, coffin or other thing" (Claire Soper, pers. comm)
Parkgrove Crematorium in Douglasmuir produced a policy statement in relation to the cremation of babies and infants reflecting these recommendations. It includes the following paragraph:
" Whilst our employees might use the terms 'ashes' and 'cremated remains' we deem these to be one and the same and defined as 'all that is left in the cremator at the end of the cremation process and following the removal of any metal'. There might be a small number of cases where there are no ashes remaining at the end of the cremation process. If this is the case our staff will contact the Applicant for Cremation and advise them of this" (Brown, 2015).
The policy statement also makes reference to the means by which Parkgrove maximise the recovery of ashes during cremation, shared cremations, disposal of ashes and record keeping, which are in line with the recommendations of Lord Bonomy.
Also as a result of these recommendations, in March 2016, the Federation of Burial and Cremation Authorities ( FBCA), the Institute of Cemetery and Crematorium Management ( ICCM), the Cremation Society of Great Britain ( CSGB) and the Association of Private Crematoria and Cemeteries ( APCC) issued a joint policy statement for infant cremation in England and Wales which re-iterated the above definitions:
"In line with Lord Bonomy's recommendations (INSERT CREMATORIUM NAME HERE) Crematorium considers cremated remains and ashes to be one and the same thing and supports the commission's definition that ashes are "all that is left in the cremator at the end of the cremation process, and following the removal of any metal". ( FBCA, 2016).
It would seem, therefore, that there is no longer room for any confusion regarding the definitions of cremated remains and ashes, and the advice that should be given to bereaved parents in terms of what is retrievable at the end of the cremation process.
6.2 The Effects of Cremation on the Human Body
When the body is subjected to extreme heat it will undergo a number of predictable changes; the skin will harden and split, the subcutaneous fat and muscle will burn, there will be dehydration and oxidation of the organic component of the body (including the organic component of bone) and eventually, at temperatures in excess of around 1000 C°, there will be re-crystallisation of the mineral component of the bone (Holden et al., 1995; DeHaan and Nurbakhsh, 2001; McKinley, 1994; Shipman et al., 1984).
As bone is heated, proteins will undergo a process of denaturation. The water that is found in the organic component of bone is removed at between 300 and 500 C° (Harsanyi, 1993 in Fairgrieve, 2008: 138). At temperatures above 700 C° the water contained within the mineral component of bone is also lost and Calcium Oxide (CaO) is formed. It has been suggested that the formation of CaO is linked to skeletal maturity ( ibid).
It is important to note that once complete combustion of the organic component of the bone has occurred, the amount of DNA present is much reduced if not lost completely. Current standard DNA analysis techniques (eg. STR analysis of nuclear DNA or mitochondrial DNA analysis) used to obtain DNA profiles from unburnt or charred remains have had very limited success when applied to calcined bone, therefore positive identification of the deceased following complete cremation is generally not possible (McDonald, pers. comm.)
Exposure to extreme heat will cause visible changes to bone and, at sufficiently high temperatures, alteration of its microstructure. In laboratory conditions it has been proven that the colour of bone changes progressively and predictably as it is heated. These colour changes range from pale yellow, through to red /brown, black, blue, grey and finally white, when all the organic matter has combusted and the bone is calcined (Shipman et al, 1984; Holck, 1986; Holden et al., 1995).
Studies at both macroscopic and microscopic levels generally agree that under conditions of extreme heat bone shrinks, splits and cracks. There is a wide variation in the degree of shrinkage reported in different studies, with figures ranging from 2 to 25% reduction from the original fresh bone (Nelson, 1992). In the experimentally controlled cases reviewed by Nelson the amount of shrinkage was found to be at the lower end of that range averaging between 3 and 5% ( ibid). A study which closely mimicked conditions in a modern crematorium involved the cremation of one half of each of five cadavers in a gas oven with a temperature range of 600 to 1000° C (Dokladal, 1971 in Mayne Correia, 1997: 227). From measurements taken on the preserved unburned half compared to the cremated half in the same individual the researcher established shrinkage rates of between 5 to 12% ( ibid).
It has been claimed that as foetal and infant bones contain more water and organic material in the form of proteins than mature bones they shrink more than the bones of adults (Vaughan, 1981 in Smith et al, 2011). Modern clinical advice corroborates the difference in body water content up to the age of 12 months, stating that it is 70% at birth, dropping to 61% at 12 months "which is about equal to the adult percentage" (Doyle, 2016: 3). Hermann (1977) found that the decrease in bone volume which occurs during cremation was greater in neonates and infants than adults where the percentage reduction never exceeded 13% (Uytterschaut, 1993) whilst in her study, Uytterschaut found that the bones of neonates and infants contract by an average of 10% ( ibid). The degree to which shrinkage occurs in neonatal and infant bones is important within the context of this investigation as it has the potential to affect the recognition of their skeletal remains following cremation.
Numerous studies have been undertaken examining the fractures which occur as a result of thermal damage to bone (Goncalves et al., 2011; Schmidt and Symes, 2008; Bontrager and Nawrocki, 2008; Buikstra and Swegle, 1989). The majority of experimental studies have shown that burning fleshed bone, as in a modern cremation, typically produces characteristic curved, transverse, thumbnail, and step fractures, deep longitudinal fractures and warping of the bone (Ubelaker, 1989; Bontrager and Nawrocki, 2008; Buikstra and Swegle, 1989). These fractures are easily distinguishable from the fractures caused by mechanical damage following cremation, although they can actually pre-dispose the bone to this type of damage. Some examples of heat induced fractures can be seen on the foetal bones in the original photographs of the foetal and neonatal remains from Seafield and Warriston.
7. The Survival of Foetal, Neonatal and Infant Skeletal Remains During and After Cremation
On the 23rd March 2016 I received information from Claire Soper relating to fourteen crematoria that had been investigated as part of the National Investigation. It identified six crematoria where cases included in the investigation had been recorded of "no remains" following the cremation of neonates and infants. These are cases referred to the National Investigation by families and do not therefore represent the oldest babies recorded as "no remains" in the whole of the registers at the crematoria. The crematoria and the ages of the individuals who had been cremated are outlined in Table Seven, below. It should be noted that in most crematoria the term 'no remains' was never used when registers were completed manually, and this term was only adopted with the advent of computerised records. So, although there may have been no remains for older babies they were recorded as 'dispersed in the Garden of Rest'.
Table Seven: Crematoria with "No Remains" Recorded (Data provided by Claire Soper)
| Crematorium |
Age of infant |
|---|---|
| Kirkcaldy |
3 hours 15 minutes |
| Linn |
9 months 3 weeks [4] |
| Daldowie |
5 months |
| Craigton |
9 days |
| Aberdeen |
5 ½ months |
| Mortonhall |
1 day |
Details of research and scientific findings are given below which provide evidence that the remains of infants of an equivalent age to those listed above (and younger), purported to have no remains, would survive cremation.
7.1 The Impact of Cremation on the Foetus, Neonate and Infant
In terms of bone and tooth survival, cancellous bone will shrink but generally retain its shape, whereas compact bone will shatter into small pieces, and un-erupted teeth and roots survive while the exposed crowns break apart (Mayne Corriea, 1997:278). The survival of bones and teeth, including those of foetuses, neonates and infants, is well documented in archaeological cremation burials of up to c. 4000 years old, even where the remains are calcined, completely mineralised and brittle (Hillson, 2009; McKinley, 1994, 1996; Downes and McGregor, 1995;
Roberts, 1995, 1998, 2001; McSweeney, 1995; Davies and Mates, 2005; Smith et al., 2011). It has also been proven through archaeological and modern crematoria studies that certain bones are more likely to survive than others and, in summary, the denser bones and those well embedded in muscle tissue are found to be most resilient (Mayne Corriea, 1997:278). This was also identified by Smith et al (2011) in their study of archaeological remains from Carthage, who observed that size differences between burnt and non- burnt bone varied for different elements, with shrinkage being more pronounced in younger individuals.
Where ossification has not begun or is in its very early stages, the cartilage or connective tissue prototype for the bone can be lost entirely in the cremation process as all the organic matter in the body is combusted. Once the bone has started to ossify, however, it will undergo broadly the same changes as adult bone during the cremation processes. That said there are some differences to take into consideration which relate to the development and maturity of the bone. It has already been noted that neonatal and infant bone loses more volume than adult bone when burnt and some studies found there was a greater degree of shrinkage in foetal bone. Fairgrieve (2008: 138) stated that neonatal bones will burn "more completely" than adult bones and less mineral residue will be left following cremation, due to a lack of Calcium Oxide (CaO) in the bones of young individuals as the intermolecular cross-links between the collagen chains have not yet developed.
It is true for adults that bone mineral density and the weight of cremated bone is affected by age, sex, stature, diet, activity and even geographical location (Van Deest et al., 2011). It follows that some of these criteria would also apply to foetal, neonatal and infant skeletons with more emphasis on the maternal environment in the former (see also Section 5.3). Some foetuses and neonates may be smaller than usual or have delayed development for their gestational age and therefore there bones may be more susceptible to damage from the heat and post-cremation mechanical damage.
In terms of gross anatomy, neonate and infant bones are thinner, smaller, less robust and lighter than adult bones therefore they will combust more quickly and at lower temperatures. It has been noted that for an adult the whole cremation process takes on average 90 minutes at a temperature of 1000º C or more, whilst cremation of an infant or foetus can be completed in 40 to 60 minutes at temperatures of 700º C (Dunlop, 2004). In the same paper, Dunlop noted that foetal skeletal remains (he does not state gestation period) could be "discerned quite clearly" following cremation at Hull Crematorium ( ibid). This is discussed further in Sections 7.2 and 7.3.
7.2 The Appearance and Size of Cremated Foetal, Neonate and Infant Remains
Evidence that foetal skeletal remains can survive modern cremation processes and are recognisable from as early as 17 weeks gestation, was obtained from two private crematoria currently operating in Edinburgh, Seafield and Warriston. Photographs of these remains were not included can be found in the report of Dame Elish Angiolini, 2014 (The City of Edinburgh Council 2014) for reasons of sensitivity but were made available on request following publication of the Report. Additional information and images supplementing the findings from Seafield and Warriston has since been obtained from Parkgrove Crematorium in Douglasmuir (Soper pers. Comm. 2015; 2016).
As explained in Technical Note One of this report, a decision was made not to display the actual photographs of the remains. Instead an artist's impression was created by the Royal Military Police Multimedia and Evidential Imagery Team, using Adobe Photoshop CS5 and a Wacom Tablet. These are shown below and following each image there is a list of the bones which are identifiable in that picture. The photographs from Parkgrove Crematorium were slightly less clear than those from Seafield and Warriston therefore the individual bones are slightly less well defined.
Image One: Seafield, Full Term Stillborn

Bones that can be identified in Image One include the femur, humerus, fibula, tibia, pelvis (ilium), scapula, possibly an ulna, multiple vertebrae (body and neural arch), a minimum number of 10 ribs, metacarpals / metatarsals, phalanges.
Image Two: Warriston, 22 Weeks Gestation
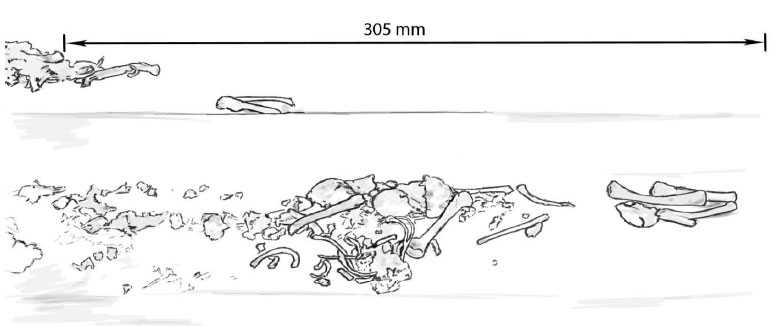
Bones that can be identified in Image Two include the femur, tibia, fibula, ilium, humerus, possible mandible and tooth crowns, metacarpal / metatarsals, a minimum number of 10 ribs.
Image Three: Parkgrove 27 weeks
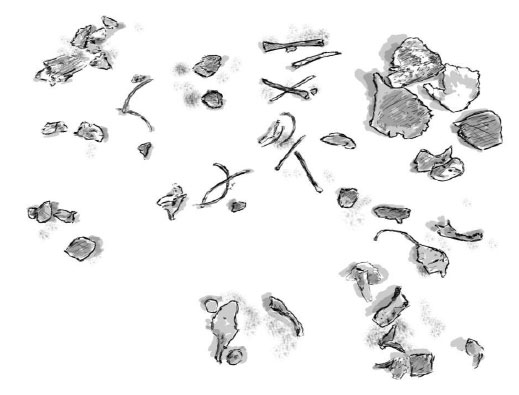
Bones that can be identified on Image Three include cranium (frontal and parietal), right and left mandible, femur, probable humerus, fibula, a minimum number of 7 ribs.
Image Four: Parkgrove 9 months Gestation
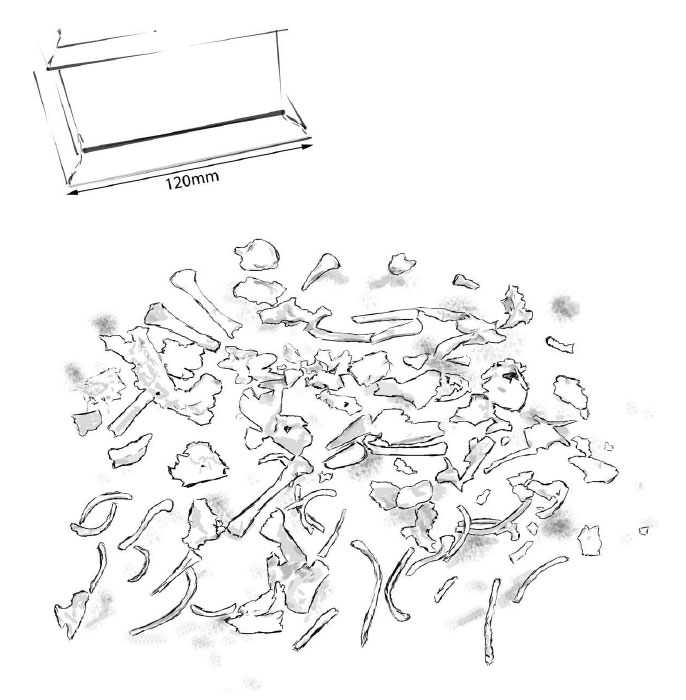
Bones that can be identified on Image Four include cranium (cranial vault and pars basilaris of occipital), mandible, clavicle, scapula, femur, humerus, tibia, fibula, possible radius, possible calcaneus (heel bone), a vertebra, a minimum number of 19 ribs.
The reproduced images above were selected on the basis that they illustrate a range of ages from a number of different crematoria where foetal and neonatal bones are clearly distinguishable post-cremation. The inference that can be drawn from this is that if remains from foetuses as young as 22 weeks in utero can be clearly distinguished then the bones of infants up to two years should be easily identifiable.
Metric data relating to the sizes of unburnt and burnt bones of infants aged 40 weeks to two years, which supplement the visual representations, can be found in Table Eight below. It presents metric data taken from the neonatal remains at Seafield, Warriston and Parkgrove and compares it to published reference data from neonates of approximately the same age (the full-term stillborn remains from Warriston referred to in the Table Seven are not shown in the drawings above). The original photographs on which the drawings were based were taken at different scales and so in order to take comparable measurements from them they were reproduced at the same scale using a number of different methods.
The images from Seafield and Warriston were reproduced with reference to a ruler which appeared in the original photograph of the cremated foetal remains at 22 weeks gestation from Warriston. The ruler was used to measure the fixed distances between the metal grooves visible on the cremation tray in the images of the full term remains from Seafield and Warriston and these measurements were extrapolated so that the photographs could be reproduced at a scale of 1:1. In the image of the 9 month gestation neonate from Parkgrove, the base of the coffin was used as a known measured distance (provided to me by Claire Soper) in order that the photograph could be reproduced at a scale of 1:1. It was then possible to take measurements of the identifiable bones in all the enlarged photographs using a standard ruler.
It should be emphasised, that the measurements taken from the photographs are approximate and that in some instances the position of the bones, for e.g. if they were placed at an angle or not lying flat, may have slightly reduced the accuracy of the measurement. Where obvious distortion could be seen, caused either by thermal damage (warping and cracking), or the angle of the photograph or bone, the measurement was not recorded and the corresponding data field was left blank.
With regard to the age and sex of the neonates from whom the data in Table Eight was derived, the sex of those from the crematoria and all the published reference data, except Trotter and Pearson, is unknown. The term "neonate" has been used for all of the babies as their ages were all described differently as follows; "full term stillborn" (Seafield and Warriston), "9 months gestation" (Parkgrove), 40 weeks (Fazekas and Kosa), "perinatal" (Trotter and Pearson).
Table Eight: Measurements of bones taken from rectified 1:1 scale photographs of full-term cremated neonates from Seafield ( SE), Warriston ( WA) and Parkgrove ( PG) Crematoria, compared to published reference data of unburnt bone.
| Skeletal Element |
Maximum Length (mm) |
||||
|---|---|---|---|---|---|
| WA |
SE |
PG |
Published Reference Data |
||
| Clavicle |
36 |
44.1 |
(Fazekas and Kosa, 1978) |
||
| Humerus |
53.5 |
54 |
64.9 |
(Fazekas and Kosa, 1978 |
|
| 65.2 (m) |
(Trotter and Pearson, 1969) |
||||
| 61.2 (f) |
(Trotter and Pearson,1969) |
||||
| Pelvis (iliac length) |
35 |
28.5 |
34.5 |
(Fazekas and Kosa, 1978) |
|
| 31.5 |
(Appuzzio et al, 1992) |
||||
| Femur |
71 |
59 |
74.4 |
(Fazekas and Kosa, 1978) |
|
| 75.4 (m) |
(Trotter and Pearson, 1969) |
||||
| 70.7 (f) |
(Trotter and Pearson, 1969) |
||||
| Tibia |
65 |
65.2 |
(Fazekas and Kosa, 1978) |
||
| 66.8 (m) |
(Trotter and Pearson, 1969) |
||||
| 60.8 (f) |
(Trotter and Pearson, 1969) |
||||
| Occipital Bone ( Pars Basilaris) |
10 |
13.1 |
(Fazekas and Kosa, 1978) |
||
| Rib* |
59 |
56 |
61.6 |
(Fazekas and Kosa, 1978) |
|
*6th rib selected as typical rib from Fazekas and Kosa reference data. Not possible to accurately determine middle rib numbers accurately from Parkgrove photograph therefore a typical rib (not the 1st, 2nd, 3rd, 10th, 11th or 12th) was selected.
It can be seen from the table above that the measurements of the bones from Warriston were broadly comparable with the published reference data. At Seafield and Parkgrove, however, the measurements were consistently shorter, most noticeably in the humerus and femur. These results have not been statistically analysed and the sample size is small, so it is difficult to interpret the findings. They could be a true reflection of the pre-cremation smaller size of the foetuses or they could indicate that a greater degree of shrinkage took place during cremation, at least in the remains from Seafield and Parkgrove.
Harsanyi (1993) reported that the diaphyses of long bones of infants and foetuses contracted by some 10% in length after cremation, while Muller et al. (1952) reported that even more shrinkage occurred in the youngest individuals (Smith et al. 2011). If shrinkage was responsible for the shorter length of the bones at Seafield and Parkgrove it did not have a detrimental effect on the preservation of the bones in question, as they appear from the images to be in a good state of preservation, and recognisable, with minimal fracturing caused by thermal or mechanical damage. If the explanation is a pre-cremation smaller size, it could be an indication that the foetus was small for its gestational age or it may even have died in utero some time before the spontaneous abortion or stillbirth occurred.
The above analyses within the context of the National Cremation Investigation provides direct, visual evidence that multiple individual skeletal elements can be recognised following cremation in individuals as young as 22 weeks. Visual evidence for the survival of skeletal remains in a cremated foetus aged 17 weeks from Seafield Crematorium was presented in my report for the Mortonhall Investigation (The City of Edinburgh Council, 2014) and reference to this was made in Lord Bonomy's recommendations (The Scottish Government, 2014; 2015). By comparing the metric data to a documented reference collection it can also be seen that in the majority of instances, if cremation is conducted carefully, there is little alteration to the size and shape of the foetal and neonatal bones.
7.3 Percentage of Bone Ash Remaining following Cremation
Experimental research has been undertaken to quantify the percentage of bone (bone ash or calcined bone) remaining in human skeletons following cremation. Trotter and Hixon (1974) studied skeletons from an early foetal period through to old age. This included 124 male and female foetuses of American Caucasoid and Negroid ancestry, which ranged in age from 16 to 44 weeks gestational age. It was possible to record the ash in even the youngest and lightest skeletons, the lightest being a white male of 16 weeks gestation which weighed 3.4 g pre- cremation. Individual percentage ash weights ranged from 58%, a white female, to 72.3% a white male (Trotter and Hixon, 1974: 13). The mean percentage ash weights showed a slight, but significant increase with age, but no statistically significant differences were found with regard to sex and ancestry ( ibid). Trotter and Hixon found that during the foetal period, the percentage ash weight increased slightly in the total skeleton and in some bones, but there was no significant trend thereafter. In the context of this investigation, this would mean that we could expect to see the same ratio of bone ash weight to total weight of skeleton in neonates and infants up to two years of age and it follows logically then, that there would be more ash remaining from a two year old infant than there would be from a neonate.
Although Trotter and Hixon removed any soft tissue from their subjects before cremation, their results for adults were comparable to the findings in research conducted by Bass and Jantz (2004) [5] on fresh cadavers in modern crematoria. The study by Trotter and Hixon is important because it illustrates that even at 16 weeks gestational age there will be survival of calcined bones or "ashes" following cremation and that this amount will increase proportionally with age.
To further quantify the total ash mass that would be present following the cremation of neonates and infants up to two years of age, data from Tables Nine and Ten were entered into an Ash Calculator. The values shown in the Table Nine were derived from World Health Organisation statistics and are shown as medians due to the fact that their data are expressed as percentiles (World Health Organisation, 2016). The information in Table Ten was provided to me by Mr Silvano (Tom) Amato, Director at FA Albins and Sons. [6]
The Ash Calculator was designed in Microsoft Excel by Intertek, a UKAS accredited testing company, on behalf of the Funeral Furnishing Manufacturing Association ( FFMA) to determine the exact volumes of ash created during cremations. It was designed and built based on research carried out on adults not children.Its purpose is to give an estimate of ash volume after the process of cremation. Its functionality caters for:-1) the differing styles of traditional, eco, and alternative coffins. 2) the differing sizes of adults and coffin sizes [7]
This work was commissioned as a result of concerns about the trend in "eco/alternative coffins" which produce more ash than a standard coffin (Crampton, 2015). It should be taken into account that there is no facility for coffins made of MDF in the Ash Calculator therefore the ash content of chipboard (1.7%) was used in the equations. This means that for neonates and infants the coffin ash mass and the total ash mass are approximate values. It should also be noted that where an infant age fell at the beginning or end of the age range given for coffin size (see Table Ten), the following weights were used in the Ash Calculator: 3 months = 0-3 month size coffin; 12 months = 3-12 month size coffin; 18 months = 12-18 month size coffin; 24 months = 18-24 month size coffin.
Table Nine: Weights and Lengths of Neonates and Infants ( WHO, 2016).
| Age (months) |
Weight (kg) |
Length (cm) |
||
|---|---|---|---|---|
| Male |
Female |
Male |
Female |
|
| 0 |
3.3 |
3.2 |
49.9 |
49.1 |
| 3 |
6.4 |
5.8 |
61.4 |
59.8 |
| 6 |
7.9 |
7.3 |
67.6 |
65.7 |
| 12 |
9.6 |
8.9 |
75.7 |
74 |
| 18 |
10.9 |
10.2 |
82.3 |
80.7 |
| 24 |
12.2 |
11.5 |
87.8 |
86.4 |
Table Ten: Coffin Sizes and Weights (see footnote 5)
| Coffin Weight (kg) |
Coffin Length (cm)* |
Age Range of Occupant (months) |
|---|---|---|
| 4 |
61 |
0-3 |
| 8 |
76 |
3-12 |
| 11 |
91.4 |
12-18 |
| 13 |
109.7 |
18-24 |
Table Eleven: Results from Ash Calculator (presented in grammes rather than kilogrammes)
| Age |
Sex |
Body Mass |
Ash (g) |
Coffin Mass | Ash (g) |
Total Mass | Ash (g) |
Vol Ash Produced (cu cm) |
|---|---|---|---|---|---|---|---|---|
| 0 |
Male Female |
184 182 |
68 68 |
252 250 |
230.75 228.67 |
|||
| 3 |
Male Female |
325 305 |
68 68 |
393 373 |
359.40 341.68 |
|||
| 6 |
Male Female |
405 379 |
136 136 |
541 515 |
494.89 470.93 |
|||
| 12 |
Male Female |
510 482 |
136 136 |
646 618 |
590.71 565.59 |
|||
| 18 |
Male Female |
601 560 |
187 187 |
788 747 |
721.29 682.93 |
|||
| 24 |
Male Female |
686 659 |
221 221 |
907 880 |
829.58 805.45 |
It can be seen from Table Eleven that in all cases the total amount of ash produced would be measurable and visible in terms of both weight and the amount of space it would occupy. The remains of a cremated six month old infant would, for example, weigh on average between 51 and 54g which for reference purposes is approximately equivalent to a pound of flour. It has also been demonstrated in Section 7.2 that the bones would still be easily recognisable even in the lowest weights.
8. The Relationship between Methods of Cremation, Survivability of Remains and Recovery of Ashes
The section below has been taken directly from the report I produced for Dame Elish and the Mortonhall Investigation (The City of Edinburgh Council, 2014). I have reproduced the text here with appropriate amendments as it equally applies to the remains of neonates and infants which should, as previously discussed, be even more evident following cremation than foetal remains.
It has been demonstrated that foetal remains of 17 weeks gestation and older can and do survive complete combustion (report produced for Mortonhall Investigation). It is also apparent from the literature and examination of the images from Seafield, Warriston and Parkgrove Crematoria that individual bones are identifiable to skeletal element from this age. If that is the case, then other explanations must be sought for the stated absence of ashes in individuals aged 40 weeks to 2 years of age. Possible explanations include:
1. The ashes have survived cremation but they have been destroyed during the recovery process or transported to the secondary chamber from where they are not recovered
2. The ashes have survived the cremation and recovery processes but human neonate or infant remains contained within them have not been identified
8.1 The ashes have survived the cremation process but they have been destroyed during the recovery process or transported to the secondary chamber from where they are not recovered
In terms of ashes being transported to the secondary chamber, details relating to this can be found in the expert report of Dr Clive T Chamberlain (The City of Edinburgh Council, 2014; The Scottish Government, 2014; 2015). The aspects of cremation which are most detrimental to neonate and infant remains appear to be the jets of air introduced into the cremation chamber and direct heat in excess of 1000° C from support burners (Dunlop, 2004). Whereas the weight of adult bones ensures that they are not carried out of the cremation chamber into the secondary combustion chamber, neonate and young infant bones are much lighter and so they may be carried through. Ashes are removed from the cremation chamber so if the lighter remains have been blown into the combustion chamber then they will not be retrievable. Clearly a less vigorous method of cremation would be of benefit when dealing with neonate and young infant remains. Lower temperatures of around 600 to 700° C are recommended by both Dr Chamberlain and Dr Dunlop, a Medical referee at Hull Crematorium. Dunlop also recommends that "no forced air is turned on" (2004: 341) and that the coffin containing the foetus / young infant is placed in a preheated furnace in a corrugated metal tray with sides [8] . Dr Chamberlain refers to modified practices at Seafield Crematoria and trays such as those described by Dunlop can be seen in Images One and Two of this report.
In terms of the process of recovery of ashes, recovery of neonate and infant ashes is closely linked to the issue of how the remains are contained during cremation. Clearly there is going to be a better chance of recovering all the small bones if they are kept together in a small metal tray which restricts dispersal during cremation. The other area of concern is how the ashes are removed once the cremation is complete. As previously discussed bones become more brittle and fragile once the organic component has been combusted and therefore they are more susceptible to mechanical damage. Usual practice is for the ashes to be raked out of the cremation chamber once they have cooled down (Bass and Jantz, 2004; Chamberlain, 2013). This process however, is extremely detrimental to delicate neonate and infant bones which may already be fractured due to thermal damage. Further fragmentation in combination with their already small size, could lead to destruction of the bone or loss amongst any accompanying burnt material. A better means of recovery of neonatal and infant remains would be to lift them out on a small tray once it has cooled down and then retrieve the bones by hand.
8.2 The ashes have survived the cremation and recovery process but human foetal, neonatal or infant remains contained within them have not been identified
The bony parts of the neonatal and very young infant skeleton might not necessarily be recognisable as skeletal remains to the untrained eye or inexperienced member of staff. At birth there are usually approximately 450 centres of ossification; the bony "pieces" of the skeleton (White and Folkens, 2005: 47) and whilst some skeletal elements such as the long bones, cranium and ribs are relatively easy to recognise, others such as the incomplete vertebrae, the tarsal bones and any newly developed epiphyses could be confused with other burnt debris. In older infants whilst some of the centres of ossification will have started to fuse, new ones will also have appeared so there will still be a large number of individual bones present that do not yet resemble their adult form. Despite this, a significant proportion of the bones will be recognisable in the older infants, they will be larger than those of the neonate and as such, even untrained staff should be able to distinguish them from coffin and other debris.
As with cremated foetal remains it may be difficult to recognise all the components of the neonatal skeleton therefore there is still a potential risk that crematoria staff might inspect the contents of the cremation chamber and wrongly conclude that there are no bones surviving. This should not happen with the infants, particularly those at the upper end of the range being considered. If there are still members of staff involved in the cremation of juveniles who have not had awareness training specifically in the appearance of cremated neonatal and infant remains, then this needs to be addressed as a matter of priority.
9 Distinguishing between Adult, Neonatal and Infant Remains that were Cremated Together
Section 5 describes the development of the human skeleton from ossification in utero to development and closure of the secondary centres of ossification. Sections 6 and 7 provide detailed information on the changes caused to the body by the cremation process and the appearance of cremated neonatal and infant bone. The information provided in these sections includes detail relating to the differences in the appearance and size of neonate, infant and adult bones. These differences are retained and still evident following cremation even if the bones become highly fragmented due to subsequent mechanical damage. Key differences are as follows:
- The cortical bone of infant and neonate remains will be far thinner than that of adults and instead of smooth joint surfaces at the ends of the bones (or fragments of) there will be undulating, unfused epiphyseal surfaces which are entirely different in appearance.
- In the infant and neonate, the major long bones such as the femur and humerus will have the same diaphyseal shape as in the adult, but they will be missing their ends, the epiphyses (for e.g. the ball part of the ball and socket joint, see Image Five).
- In the neonate and infant there will be multiple small bones, such as vertebrae and developing pelvic bones, which are still in "component parts" and as such, bear little resemblance to their adult form (See Image 5).
Image Five: a) Reference casts of bones from an infant aged 1-2 years, includes unfused bones of the right pelvis (encircled in red), humerus, femur (on right of picture) and clavicle; b) reference cast of adult right side of pelvis; fully formed and development completed; c) reference cast of adult femur, fully formed and development completed.
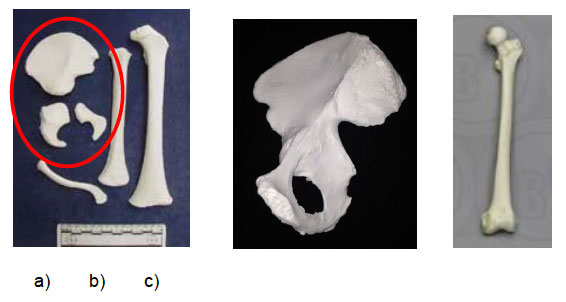
Source of Images: a) and b) France Casting ( http://www.francecasts.com ), c) Bone Clones ( https://boneclones.com)
It is also possible to distinguish between infant and adult teeth and in the neonate and infant there will be deciduous dentition present that will not be there in the adult. If these have erupted they may not survive the cremation process but if they are un-erupted within the jaw, they may well be preserved. If the infant is slightly older there may also be developing permanent dentition in the jaws, which will look different from erupted adult dentition which has completed its growth and development.
The differences described above can be used to determine whether infant remains have been cremated together with those of an adult, even if they have become commingled and fragmented as a result of the jets of air and direct heat in the cremation chamber. In addition to this, if more than one individual was cremated at the same time, regardless of whether they were adult or infant, there would be repeated skeletal elements, for example two right femurs or two left clavicles (or parts of). If this was the case, then it could be stated with confidence that at least two individuals had been present at the time of cremation. This could be applied to any of the identifiable bones in the skeleton, although it would not be sensible to try and count multiple bones such as ribs or vertebrae, rather than bones which are paired.
10. Conclusions
I was asked to provide information and commentary on the following for the purposes of this report:
- The skeletal development of infants aged 40 weeks to 24 months.
- The size and appearance of the remains of infants within the above age range
- The amount of ash that would be produced from the cremation of those infants
- Whether it would be possible to distinguish between adult and infant skeletal remains if they had been cremated together
- Whether it was possible that the skeletal remains of infants up to two years of age might not survive the cremation process.
The skeletal development of neonates and infants within the specified age range has been described in Section 5 and reference data has been provided on the average lengths of easily identifiable bones (Tables One to Six). Section 7 dealt specifically with the effects of cremation on neonatal and infant bones, and images of foetal and neonatal cremated remains from three crematoria, Warriston, Seafield and Parkgrove were reproduced for reference purposes. The metric data relating to these images was compared to reference standards in Table Eight.
Published standards on average weights and lengths of neonates and infants were entered into an Ash Calculator (provided to me by Claire Soper), together with data obtained from a Funeral Directoron average weights of coffins for infants of the specified ages. This enabled the approximate body ash mass, coffin ash mass and total ash mass to be calculated for neonates and infants aged 40 weeks, 3, 6, 12, 18 and 24 months (Table Eleven).
All of the above demonstrated unequivocally that the skeletal remains of infants of the specified ages would survive the cremation process and be identifiable. Clearly the older the child the easier it would be to distinguish human remains from extraneous material such as coffin debris, clothing and any personal effects. For example, the femur of a two year old child has a very distinctive form and an average length of 17.2 cm in the male and 17 cm in the female. The cremated remains of a child of that age would weigh approximately 906 grams for a male and 880 grams for a female, which is a considerable amount of material to overlook. Even in the youngest age group analysed, 40 weeks gestation, there would still be approximately 250 to 252 grams of ashes following cremation.
My previous report prepared for Dame Elish provided evidence that the skeletal remains of foetuses as young as 17 weeks can and do survive the cremation process (City of Edinburgh Council, 2014). Taking that into consideration alongside the data presented in this report, it is inconceivable that there would be nothing left of newborn babies and infants aged up to two years following cremation. The "no ashes" or "no remains" policies at the Crematoria of concern must therefore be related to issues surrounding recovery processes, the ability to recognize burnt skeletal remains, and / or individual or corporate management decisions. The same applies to the reasoning that the remains of infants and adults could not be distinguished and separated in instances where they had been cremated together.
It is my understanding that following the publication of the findings from the Mortonhall and Infant Cremation Commission Investigations, the Scottish Government produced some clear guidelines and recommendations which should, if followed, ensure that "no ashes" / "no remains" policies are no longer in place for newborn babies and infants up to the age of two years.
Appendix One:
Diagram of the Neonatal Skeleton (Adapted from Buikstra and Ubelaker, 1994)
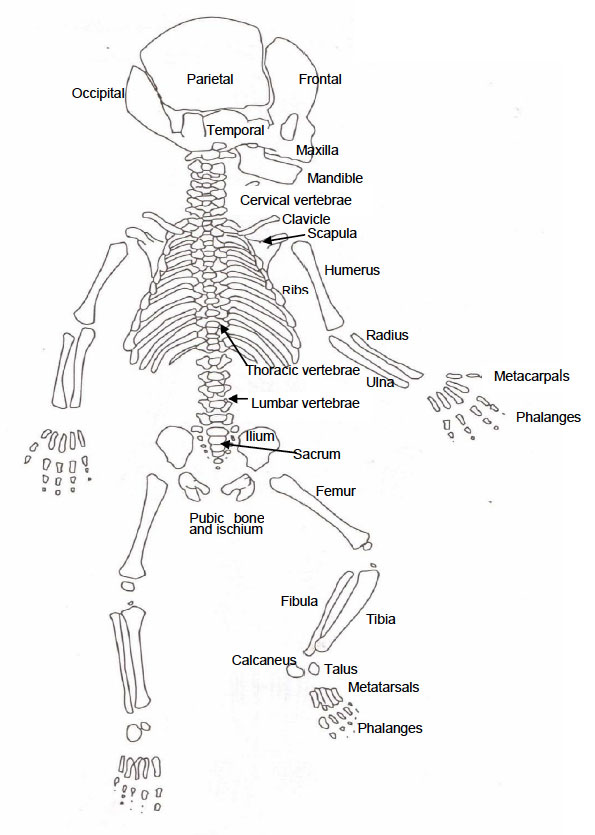
Diagram of Juvenile Skeleton (Adapted from Buikstra and Ubelaker, 1994)
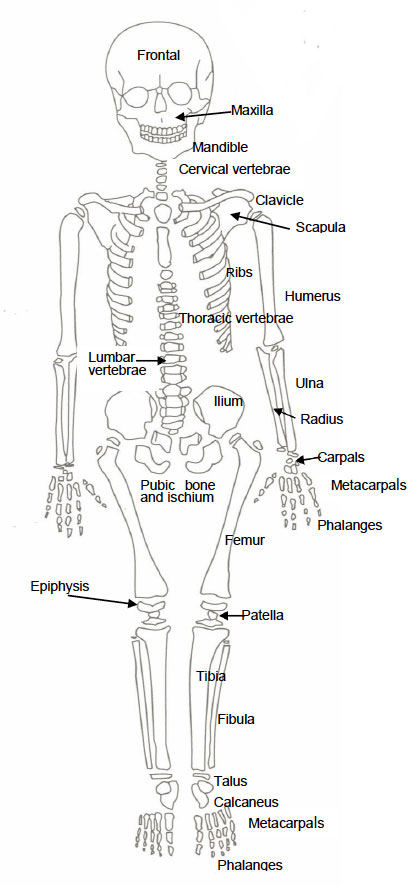
Appendix Two: Glossary of Terminology
i) Terminology Relating to Age
Embryo: The first 8 weeks of intra-uterine life
Foetus: From week 9 to birth
Perinatal: Around the time of birth, from 24 weeks gestation to 7 post-natal days
Neonatal: From birth to 28 days
Infant: From birth to 1 year
Pre-term: from < 37 weeks (258 days) gestation
Full-term: from 37-42 weeks (259-293 days) gestation
Post-term: > 42 weeks (294 days) gestation
Stillbirth: Infant born after gestational period of 24 weeks who shows no signs of life
Gestational age: The number of days or weeks that have passed since the first day of mothers last menstrual period
Conceptional age: The number of days or weeks that have passed since conception i.e. fertilization of the egg.
(Scheuer and Black, 2000, Appendix 1)
Infancy: First 3 years of life (Bogin, 1999; Black and Maat, 2010)
ii) General Terminology
Articulate(s) Adjacent to and joins with, e.g. The bottom end of the femur articulates with the top end of the tibia to form the knee joint or the base of the skull articulates with the 1st cervical vertebra of the neck.
Basi-cranium The bones of the base of the skull
Body of vertebra Main part of the vertebra that constitutes the weight-bearing portion of a vertebra
Cancellous bone/Trabecular bone Spongy, porous, lightweight bone with a honeycomb structure, found under compact bone e.g. within vertebrae, in the ends of long bones, filling short bones and sandwiched within flat bones. The spaces in cancellous bone are filled with marrow
Chondrocytes The only cells within cartilage, they produce and maintain the cartilage matrix
Collagen chains Chains of the specific amino acids which form collagen
Collagen The major protein of the white fibers of connective tissue, cartilage, and bone
Compact bone/Cortical bone Solid, dense bone found in the walls of bone shafts and on external bone surfaces including joint surfaces
Cranium Bones of the skull excluding the mandible (lower jaw)
Deciduous dentition The first set of teeth, (milk teeth)
Dentine The calcified tissue beneath the enamel in a tooth
Diaphysis Shaft of a long bone
Enamel The calcified tissue covering the outer layer of the crown of the tooth (smooth outer layer of the tooth)
Endochondral The formation of bone within a cartilage model Epiphyseal plate Area of growing tissue at the end of the metaphysis
Epiphysis The secondary centres of ossification that fuse on to the main part of the bone
Foramen magnum Large hole at the base of the skull through which the brainstem passes and turns into the spinal cord
Hydroxyapatite The calcium containing constituent of bone and teeth
Ilium Thin bladelike section of one of the two pelvic bones, the part just above the hip socket
Intermolecular cross-links The bonds between molecules
Intramembranous The formation of bone within a membrane in the absence of a cartilage model
LMP Last Menstrual Period
Mesenchymal Referring to the mesenchyme or mesenchymal tissue
Mesenchyme Meshwork of embryonic connective tissue in the mesoderm (the middle of the three cell layers of the developing embryo) from which are formed the connective tissues of the body (including cartilage and bone) as well as blood and the lymphatic vessels
Metacarpals Long bones of the hand, between the wrist and the fingers
Metaphysis The expanded, flared ends of long bones, adjacent to the cartilage growth plate and epiphysis
Metatarsals Long bones of the mid-foot
Neural arch of vertebra The part of the vertebra which forms the arch behind the body enclosing the spinal cord in life
Occlusal cap The structure of enamel and dentine when the crown is complete prior to the formation of the root of the tooth
Ossification centre The site where bone begins to form within the membrane or cartilaginous model as a result of the accumulation of osteoblasts in the connective tissue.
- Primary the first site where bone begins to form in the shaft of a long bone or in the body of an irregular bone
- Secondary centre of bone formation appearing later than a primary centre, usually in an epiphysis
Ossification The process of bone formation
Osteoblast A cell from which bone develops; a bone-forming cell
Osteoclasts A type of bone cell which resorbs bone during bone remodelling and shaping
Pars basilaris Base part of a bone, in the occipital bone it is the thick, square projection in front of the foramen magnum
Pars lateralis Lateral part of a bone, in the occipital bone, the parts which lie either side of the foramen magnum and articulate with the temporal bones
Phalanges Finger or toe bones (see Appendix One)
Tarsals Seven irregular shaped bones which articulate together between the lower leg bones and the metatarsals to form the ankle and posterior foot (calcaneus and talus are shown in appendix one)
Appendix Three: Reference sources, measurements of Neonatal and infant bones
Fazekas and Kosa, 1978, in Scheuer and Black, 2000, pages 61, 147, 243, 250, 270, 288, 373, 393, 414.
Frontal bone, femur, humerus, tibia, clavicle, scapula, pelvis, occipital bone, rib. Reference data for neonate. The study used 138 spontaneously aborted white European foetuses. The sex of the foetuses is not stipulated, measurements were taken from dry bone. Age was taken from maternal history and it is not specified whether it was given in gestational or conceptual weeks.
Yarkoni et al, 1985 in Scheuer and Black, 2000 page 250
Clavicle. Reference data for neonate. Measurements taken from ultrasound scans
Trotter and Peterson, 1969 In Scheuer and Black, 2000, pages 288, 394, 415 Femur, humerus, tibia. Reference data for perinate
Apuzzio et al., 1992 in Scheuer and Black, 2000 page 373 Pelvis. Reference data for neonate taken from ultrasound scans.
Gindhart 1973 in Scheuer and Black, 2000, page 415
Tibia. Reference data for infants aged 3 to 24 months. Males and females, diaphyseal length only, measurements taken from radiographs
Hrdlicka, 1942 in in Scheuer and Black, 2000, page 271 Scapula. Reference data for neonate
Maresh, 1970 in Scheuer and Black, 2000, pages 289, 394
Femur, humerus, tibia. Reference data for infants aged 3 to 24 months. Measurements taken from bone, diaphyseal length only, males and females
Young 1957 in Scheuer and Black, 2000, page 108
Frontal Bone. Reference data for infants aged 3 to 24 months (excluding 18 months)
Black and Scheuer, 1996, in Scheuer and Black, 2000, page 252
Clavicle. Reference data for infants aged 3 to 24 months. Sex and sample size not stipulated. Ages are given in ranges of 6 month intervals, e.g. birth to 6 months, 7 months to 1 year, 1 year to 1.5 years rather than single value ages. In order that the data could be used in this report, the bottom value in the range was cited
Saunders, 1993 in Scheuer and Black, 2000, page 271
Scapula. Reference data for infants aged 6 to 24 months (excluding 18 months). The same method was applied to data as described above (with ref to Scheuer and Black 1996) as these data were also given in ranges
Vallois, 1946 in Scheuer and Black, 2000, page 271 Scapula. Reference data for neonate and 24 month infant
Appendix Four: References
Ameen, S., Staub, L., Ulrich, S., Vock, P., Ballmer, F. and Anderson, S.E., 2005. Harris lines of the tibia across centuries: a comparison of two populations, medieval and contemporary in Central Europe. Skeletal Radiology, 34 (5), pp. 279-284
Bagnall, K.M., Harris, P.F. and Jones, P.R.M., 1982. A Radiographic Study of the Longitudinal Growth of Primary Ossification Centres in Limb and Long Bones of the Human Fetus. The Anatomical Record, 203, pp. 293-299
Bass, W. M. and Jantz, R. L., 2004. Cremation Weights in East Tennessee. Journal of Forensic Science, 49 (5), pp. 1-4
Behrents, R.G. and Broadbent, H.B., 1984. A Chronological Account of the Bolton-Brush Growth Studies: In Search of Truth for the Greater Good of Man https://dental.case.edu/media/school-of-dental-medicine/departments--programs/bolton- brush/Chronological_Account_BoltonBrush.pdf Accessed 6th April 2016
Biswas and Iqbal, 1998. Mosby's Crash Course: Musculo-Skeletal System. Mosby
Black, S. and Maat, G., 2010. Principles of Physical Age Estimation, in S. Black, A Aggrawal and J. Payne-James, eds. Age Estimation in the Living: The Practitioner's Guide. Wiley- Blackwell, pp. 77-94
Bontrager, A.B, and Nawrocki, S.P., 2008. A Taphonomic Analysis of Human Cremains from the Fox Hollow Farm Serial Homicide Site, in: C.W. Schmidt and S.A. Symes, eds. The Analysis of Burned Human Remains. Elsevier Ltd, pp. 211-226
Brown, G. 2015. Infant Cremation Policy Statement https://parkgrovecrem.wordpress.com/infant-cremation-policy-statement/ Accessed 24th March 2016
Buikstra, J.E. and Swegle, M. 1989. Bone Modification Due to Burning in R. Bonnichsen and M.H. Sorg, eds, Bone Modification, 1st Volume. University of Maine, pp. 247-258
Buikstra, J.E. and Ubelaker, D.H., 1994. Standards for Data Collection from Human Skeletal Remains. Arkansas Archaeological Survey Research Series No.44
Chamberlain, C. T., 2013 The Cremation of Foetuses, Neonatal and Infant Remains. Specialist report produced for Mort0nhall Investigation
Crompton, D. 2015. Letter to Judge Peter Thornton on behalf of the Funeral Furnishing Manufacturing Association
Davies, D.J. and Mates, L.H., 2005. Encyclopaedia of Cremation. Ashgate, pp.132-152
DeHaan, J.D. and Nurbakhsh, S., 2001. Sustained Combustion of an Animal Carcass and its Implications for the Consumption of Human Bodies in Fires. Journal of Forensic Science, 46 (5), pp. 1076-1081
De Onis, M., Garza, C., Onyango, A.W., Rolland-Cachera, M.F., le Comite de nutrition de la Societe francaise de pediatrie, 2009. WHO growth standards for infants and young children Archives de Pediatrie,16, pp47-53
Downes, J and McGregor, G. 1995. Linga Fiold, Sandwick, Orkney. Glasgow University Archaeological Research Division
Doyle, D.A, 2016. Physical Growth of Infants and Children (Professional Version) http://www.msdmanuals.com/en-gb/professional/pediatrics/growth-and-development/physical-growth-of-infants-and-children Last full review / revision December 2012, Accessed 6th April 2016
Dunlop, J. M., 2004 Cremation of Body Parts and Foetuses. Journal of Obstetrics and Gynaecology, 24 (4) pp. 341 to 342
Fairgrieve, S., 2008. Forensic cremation: Recovery and analysis. CRC, Boca Raton.
FBCA, 2016. Joint Policy Statement for Infant Cremation http://www.fbca.org.uk/ Accessed 22nd March 2016
Gonçalves, D., Thompson, T.J.U. and Cunha, E., 2011 Implications of heat-induced changes in bone on the interpretation of funerary behaviour and practice. Journal of Archaeological Science 38, pp 1308-1313
Goodman, A. H., Martin, D.L., Armelagos, G.J. and Clark, J., 1984. Indications of Stress from Bones and Teeth in M.N. Cohen and G.J. Armelagos, eds. Paleopathology at the Origins of Agriculture. Academic Press.
Harsanyi, L., 1993. Differential diagnosis of human and animal bone, in, G Grupe and A.N. Garland, eds, Histology of Ancient Human Bone. Berlin Springer-Verlag, pp.79-94
Holden, J.L., Phakey, P.P. and Clement, J.G., 1995. Scanning Electron Microscope Observations of Heat-Treated Human Bone. Forensic Science International, 74, pp. 29-45
Hillson, S., 2002. Dental Anthropology. Cambridge University Press
Hillson, S. 2009. The World's Largest Infant Cemetery and its Potential for Studying Growth and Development. Hesperia Supplements, 43, New Directions in the Skeletal Biology of Greece, pp. 137-154
Holck, P., 1986. Cremated bone: A medical and anthropological study of an archaeological material on cremation burials, PhD Thesis, Anatomical Institute, University of Oslo, 1986
Huelke, D.F., 1998. An Overview of Anatomical Considerations of Infants and Children in the Adult World of Automobile Safety Design. Annual Proceedings of the Association for the Advancement of Automotive Medicine, 42, pp. 93-113.
Kennedy, K.A.R., 1996. The Wrong Urn: Commingling of Cremains in Mortuary Practices. Journal of Forensic Sciences, 41 (4), pp. 689-692
Lobo, I. and Zhaurova, K., 2008. Birth defects: causes and statistics. Nature Education, 1(1):18
Malina, R.M., Bouchard, C., Bar-Or, O., 2004. Growth, Maturation and Physical Activity Human Kinetics.
Mayne Correia, P. M., 1997. Fire Modification of Bone: A Review of the Literature, in W.D. Haglund and M.B.Sorg, eds. Forensic Taphonomy: The Post-mortem Fate of Human Remains. CRC Press, Boca Raton, F.L. pp. 275-293
McKinley, J.I., 1994. Pyre and grave-goods in British cremation burials, have we missed something? Antiquity, 68, pp. 132-4
McKinley, J. I., 1996. Linga Fiold, Sandwick, Orkney; Human Bone Report with Discussion of Pyre Technology and Ritual. Unpublished report on behalf of GUARD, Glasgow
McSweeney, K., 1995. The Excavation of Neolithic, Bronze Age and Early Historic Features near Ratho, Edinburgh. Proceedings of the Society of Antiquaries of Scotland, 125, pp. 69-138
Mongelli, M., 2012. Evaluation of Gestation MEDSCAPE http://emedicine.medscape.com/article/259269-overview
Nelson, R., 1992. A Microscopic Comparison of Fresh and Burned Bone. Journal of Forensic Sciences, 37 (4), pp. 1055-1060
Roberts, C., and Manchester, K., 1997. The Archaeology of Disease. 2nd Edition. Sutton Publishing Ltd. Cornell University Press
Roberts, J. A., 1995. The taphonomy of cremated human remains: an experimental study of the fragmentation of bone from the bronze age cremation cemetery at Linga Fold, Orkney. Unpublished MSc dissertation, University of Sheffield
Roberts, J. A., 1998. Cremated Human Remains, in, McGregor G, The excavation of a cordoned urn at Benderloch, Argyll. Proceedings of the Society of Antiquaries of Scotland, 128, pp. 143-159
Roberts, J. A., 2001. Human Remains Report, Crantit Chambered Cairn, Orkney. Canmore Ref: HY40NW 17, http://canmore.rcahms.gov.uk/en/site/141720/details/crantit/
Sanders, J., 2009. Age Estimation of Fetal Skeletal Remains from the Forensic Context. MA Dissertation, University of Montana, Missoula.
Sawasdee, K., Tiensuwan, M., Siripinyanond, A.,Chirachariyavej, T. and Smith, S.M.,2012. Elemental analysis of burnt human bone for classifying sex and age at death by logistic regression. Analytical Methods, 2012 (4) pp 1769-1775
Scheuer, L. and Black, S., 2000. Developmental Juvenile Osteology. Academic Press, London Schmidt, C.W. and Symes, S.A, 2008. The Analysis of Burned Human Remains. Elsevier Ltd
Schutkowski, H., 1987. Sex determination of fetal and neonate skeletons by means of discriminant analysis. International Journal of Anthropology, 2 (4), pp. 347-352
Shipman, P., Foster, G. and Scheoninger, M., 1984. Burnt Bones and Teeth: an Experimental Study of Color, Morphology, Crystal Structure and Shrinkage. Journal of Archaeological Science, 11, pp. 307-325
Simpson and Kunos, 1998. A radiographic study of the development of the human mandibular dentition Journal of Human Evolution, 35, pp. 479-505
Smith, P., Avishai, G., Greene, J.A. and Stager, L.E., 2011. Aging cremated infants: the problem of sacrifice at the Tophet of Carthage. Antiquity Vol. 85 (329), pp. 859-874
The City of Edinburgh Council, 2014. Mortonhall Crematorium Report Published https:/ /www.edinburgh.gov.uk/news/article/1531/mortonhall_crematorium_report_published Updated 30th April 2014, accessed 24th March 2016
The Scottish Government, 2014 . Report of the Infant Cremation Commission http://www.gov.scot/Publications/2014/06/8342/3
The Scottish Government, 2015. Infant Cremations: National Investigation http://www.gov.scot/Topics/Health/Policy/BurialsCremation/NationalInvestigation Updated 29th September 2015, accessed 24th March 2016
Trotter, M. and Hixon, B. B., 1974. Sequential Changes in Weight, Density and Percentage Ash Weight of Human Skeletons from an Early Fetal Period through Old Age. The Anatomical Record, 179 (1) pp. 1-18
Ubelaker, D.H., 1989. Human Skeletal Remains: Excavation, Analysis, Interpretation. Taraxacum Press, Washington
Uysal, T., Ramoglu, S.I., Basciftci, F.A and Sari, Z., 2006. Chronologic age and skeletal maturation of the cervical vertebrae and hand-wrist: Is there a relationship? American Journal of Orthodontics and Dentofacial Orthopedics Vo. 130, pp. 622-628
Uytterschaut, H., 1993. Human Bone Remodelling and Aging in: G Grupe and A. N. Garland (eds) Histology of Ancient Human Bone: Methods and Diagnosis. Springer-Verlag, Berlin.
Van Deest, T.L., Murad, T.A., Bartelink, E.J., 2011. A Re-examination of Cremains Weight: Sex and Age Variation in a Northern Californian Sample. Journal of Forensic Sciences, 56 (2), pp. 344-349
White, T.D. and Folkens, P.A., 2005. The Human Bone Manual. Elsevier Academic Press, pp. 359-400
World Health Organisation, 2016. The WHO Growth Standards http://www.who.int/childgrowth/en/ Accessed 25th March 2016
Contact
There is a problem
Thanks for your feedback