Application for Bovine Spongiform Encephalopathy (BSE) negligible risk Status for Scotland: consultation
Consultation on the application to the World Organisation for Animal Health (OIE) for Bovine Spongiform Encephalopathy (BSE) negligible risk status as a region of the UK.
Chapter 1: Overview and Policy Proposal
Current BSE Controls
1.7 Due to the strong evidence of a link between variant Creutzfeldt-Jakob Disease (v CJD) and c BSE, BSE was made a notifiable disease by the UK Government in 1988. Exposure to BSE through the consumption of infected or contaminated meat is believed to be the primary cause of v CJD in humans. The outbreak of BSE in the 1980s led to the removal of older cattle from the food chain, a greater burden of regulation on the food and feed chain and the 1996 beef export ban. The escalation in the disease in cattle continued until 1993 when the effect of policy interventions made by Governments began to be visible, and the number of cases in Scotland declined steadily from that time. Scotland has been BSE-free since 2009.
1.8 The pathogen which causes BSE is most commonly found in specific tissues of infected cattle. These include the brain, spinal cord, tonsils, vertebral column, mesentery, caecum and small intestine and are referred to as Specified Risk Material ( SRM). There are strict controls in place to protect consumers, including:
- a ban on the use of processed animal protein ( PAP) in feed for farmed ruminants as a basic preventive measure against BSE transmission; and
- a requirement to remove SRM to minimise the risk of infective material entering the food chain from cattle in the early stages of BSE when disease is not otherwise apparent.
1.9 Animal feed containing BSE-infected meat and bone meal ( MBM), a product derived from the processing of dead livestock and abattoir waste including dead livestock, was recognised at an early stage of the BSE epidemic as the major source of infection responsible for the spread of BSE among cattle. A ban on the feeding of ruminant-derived MBM to ruminants was introduced in the UK in 1988 (1989 in Northern Ireland). On 31 July 1996, this ban was reinforced and extended to the feeding of mammalian MBM to all farmed animals. In 2001, the EU introduced a ban on feeding animal protein to ruminants and processed animal protein to farmed animals. These feed bans, and their enforcement, have been highly effective in controlling the incidence of c BSE in the UK and across the EU.
1.10 BSE testing of those cattle that were considered to pose a risk to human health, the feed ban and the removal and appropriate disposal of SRM are seen as the most important controls in place to protect consumers.
1.11 In July 2010, the European Commission outlined future steps regarding BSE/ TSEs in a plan known as the TSE Roadmap. [1] This initiative outlined possible amendments to TSE rules with the objective of reviewing TSE control measures to ensure that they were proportionate to the risk, while ensuring a high level of food safety. Under this Roadmap, amendments to TSE rules have been made in the light of scientific evidence from the European Food Safety Authority ( EFSA). EFSA has published opinions on risks of changing the BSE testing programme and also relating to reducing the SRM controls. [2]
1.12 As a result of amendments to the UK BSE testing programme, there is now no requirement to test healthy cattle slaughtered for human consumption for BSE. However fallen cattle (cattle that die on the farm), cattle slaughtered for welfare reasons and cattle showing signs of BSE at ante-mortem inspection, are all tested for BSE. In addition, awareness programmes, surveillance and monitoring, compulsory notification and investigation of suspects remain in place. This regime ensures that a robust programme is in place to detect any re-emergence of c BSE or the occurrence of sporadic cases.
Surveillance for BSE/ TSEs
1.13 Surveillance for TSEs is carried out in the United Kingdom in livestock susceptible to the disease, including cattle, sheep and goats. All EU Member States carry out TSE surveillance in line with EU law. The main aim is to monitor trends in disease incidence and prevalence to evaluate the effectiveness of TSE disease controls. Surveillance is not in itself protection against disease, but supports essential control measures that exclude affected animals and remove designated high risk tissues from the food chain. The surveillance data also contributes to the TSE status of each country. There are two types of surveillance:
- Passive surveillance - animals with clinical signs suspicious of BSE or scrapie are reported to an Animal and Plant Health Agency ( APHA) office and are investigated. Such cases are killed and the examination of the brain determines whether the animal was affected by BSE or scrapie. APHA has been recording and analysing data from cattle since the start of the BSE epidemic in 1986, and for scrapie in sheep and goats since this disease became notifiable in 1993.
- Active surveillance - in addition to passive surveillance the EU requires all Member States to carry out active surveillance for TSEs:
- cattle carcases have been subject to testing since July 2001; and
- sheep and goat carcases have been tested since January 2002.
1.14 As predicted by epidemiological modelling, very few cases of BSE in cattle are now seen. Following a peak of over 36,000 clinical cases in the UK in 1992, the number of new cases detected by active and passive surveillance continues to decline year on year, with just 2 cases confirmed in the UK in 2015.
Table 1: Confirmed cases of BSE by passive surveillance in Scotland 1990 to 2016 [3]
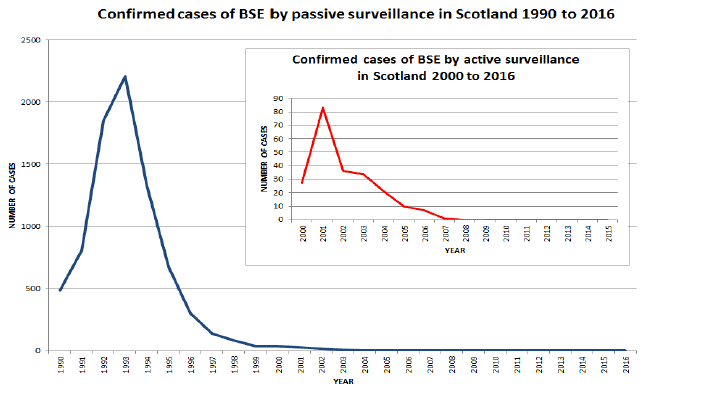
1.15 As incidence of the disease in cattle continues to fall controls across the EU are reviewed in accordance with the Commission's TSE roadmap. Whilst making proportionate reductions in testing, it is important to maintain an appropriate level of surveillance as a safety measure against any unexpected re-emergence of the disease in cattle. For example, the criteria for inclusion in the testing programme have been changed over the years in response to regular risk assessments. With falling numbers of BSE cases across the EU, the requirement to carry out TSE testing on healthy slaughtered cattle was relaxed in 2013 and now only at 'risk cattle' such as fallen stock aged over 48 months, where BSE is most likely to be detected, are routinely tested under EU law.
1.16 Of the ~180,000 cases of BSE confirmed in the UK, only 164 were born after the introduction of the July 1996 feed control. These UK cases, born after 31 July 1996, are termed BARBs (Born After the Reinforced Ban).
1.17 Following a confirmed case of c BSE in Wales in June 2015, it will be 2020 before the UK as a Member State can make an application for BSE NR Status. Although it is anticipated that England could satisfy the requirements to apply for BSE NR status in 2018, Scotland is eligible to make an application on a more rapid timetable.
Question 1
Are you in favour or against an application for BSE NR status for Scotland? Please give reasons.
Classification of BSE Risk Status - The World Health Organisation for Animal Health ( OIE)
1.18 The World Organisation for Animal Health (also known as the OIE ( www.oie.int)) is the intergovernmental organisation responsible for monitoring and improving animal health worldwide. Complying with OIE requirements is the basis of all international trade and it is this body that classifies the BSE risk status of the cattle population of a country on the basis of a risk assessment. OIE classification criteria is taken into account by the Commission in arriving at a decision on an application for categorisation. The three categories of risk for countries or regions can be summarised as follows:
- Negligible Risk ( NR) is defined as a country or region where the requisite risk analysis has been conducted, that has demonstrated that appropriate measures have been taken to manage the risks identified for the relevant period of time and that has demonstrated that there are sufficient surveillance and controls in place to meet a legislative "points target". The country or region must undertake awareness campaigns, notification and investigation and sampling. Neither meat and bone meal, nor greaves must have been fed to ruminants during the previous eight years. Further requirements depend on whether or not there have been indigenous BSE cases. If there have been, they must have occurred in an animal born at least 11 years ago, and the requirements are that BSE cases, bovine animals in contact with them and in some circumstances all bovine animals in the same herd and at-risk, must be identified, their movements restricted, and be destroyed on slaughter or death.
- Controlled Risk ( CR) countries or regions are those where the required risk analysis has been conducted, that has demonstrated that appropriate measures are being taken to manage those risks identified but that a longer period is required for their application, and that has demonstrated that the surveillance and controls are in place to meet a legislative "points target". The country or region must undertake awareness campaigns, notification and investigation, and sampling, but in this category these activities have only been ongoing for less than 7 years, and/ or the country or region must be able to demonstrate that ruminants do not eat meat-and-bone meal or greaves but in this category these bans have not yet been demonstrably ongoing for 8 years. Further requirements apply where a country or region has had an indigenous case of BSE. In these cases, all BSE cases, bovine animals in contact with them and in some circumstances all bovine animals in the same herd and at risk, must be identified, their movements restricted, and be destroyed on slaughter or death.
- Undetermined Risk ( UR) countries or regions are those whose BSE determination has not been concluded, or those not meeting the conditions applying to the other categories.
1.19 In applications for BSE risk categorisation, countries must demonstrate compliance with the provisions of the OIE Terrestrial Animal Health Code [4] -
in particular, as they apply in the following areas:
- policies designed to protect animal and human health are based on an appropriate assessment of risk;
- BSE awareness, education and reporting programs have been implemented;
- an appropriate feed ban is in place;
- there is diagnostic competency within the laboratory system; and
- BSE surveillance has been conducted in accordance with the OIE's BSE guidelines.
Differing controls on Specified Risk Material ( SRM) apply to NR and CR status
1.20 At the Standing Committee on Plant, Animals Food and Feed ( SCoPAFF) on 17 March 2015 [5] , the Commission agreed proposals to relax Specified Risk Material ( SRM) controls for Member States which have NR status. This brings EU rules more in line with the OIE requirements for non- EU countries. The change means that for Member States which have NR status:
- bovine material composed of those tissues at greatest risk of carrying potentially infective material (skull, brain, eyes and spinal cord of animals over 12 months of age) is still classified as SRM;
- all other bovine tissues would be able to enter food and feed chains; this includes the last 4 metres of the small intestine, the tonsils, vertebral column, mesentery and caecum.
. Table 2: Category and designation of bovine tissues from CR and NR countries
| Bovine Tissues |
Member States and non - EU countries with CR or UR BSE risk |
Member States with NR status |
|---|---|---|
| Skull, excluding the mandible, including the brain and eyes, of bovine animals over 12 months |
SRM |
SRM |
| Spinal cord of bovine animals over 12 months |
SRM |
SRM |
| Tonsils of all bovine animals |
SRM |
Food and feed |
| Vertebral column (backbone/spine) excluding the vertebrae of the tail, the spinous and transverse processes of the cervical, thoracic and lumbar vertebrae and the median sacral crest and wings of the sacrum, but including the dorsal root ganglia, of bovine animals over 30 months |
SRM |
Food and Feed |
| Mesentery (fold attaching the small intestine to the posterior body wall), including mesenteric fat, mesenteric ganglion complex and mesenteric nerves, of bovine animals of all ages |
SRM |
Food and Feed |
| Caecum (part of the large intestine) of all bovine animals |
SRM |
Food and Feed |
| Last 4 meters of the small intestine of bovine animals of all ages (small intestine comprises of duodenum, jejunum and ileum) |
SRM |
Food and Feed |
1.21 All SRM tissues are sent for destruction by incineration, or by rendering followed by incineration. In Scotland, as a CR region, SRM currently includes:
- in all cattle: the skull excluding the mandible but including the brains and eyes. The spinal cord of animals aged over 12 months;
- the vertebral column excluding the tail, the spinous and transverse processes of the cervical, thoracic and lumbar vertebrae and the median sacral crest and wings of the sacrum, but including the dorsal root ganglia, of cattle aged over 30 months; and
- the tonsils, the last four meters of the small intestine, the caecum and the mesentery of cattle of all ages.
1.22 It is permissible to use parts of the intestine for feed and food. However, operators have not been able to realise all of the potential benefits due to the operational difficulty in separating the mesentery from that part of the intestine which is not SRM.
Question 2
Do you have any comments in relation to the benefits from the reduction in the volume of SRM? What advantages or disadvantages do you anticipate from the reduction in the volume of SRM?
A regional application for BSE Negligible Risk status?
Criteria for making an application
1.23 Applications by Member States ( MSs) to be officially recognised as having a NR or CR BSE risk status are considered by the OIE (through the adoption of a resolution by the World Assembly of Delegates of the OIE) at the General Session in May every year. Under requirements defined by the OIE and the Commission Regulation ( EC) No 999/2001 (the EU TSE Regulation), a country or region can apply for BSE NR Status provided they have:
- a surveillance programme to detect TSEs;
- the latest date of birth of a positive classical BSE case being at least 11 years ago: and
- a control strategy in place for positive BSE cases, SRM and feed.
Current Position in Scotland
1.24 A range of food safety controls have been in place to reduce the risk to consumers across the UK since the late 1980s. These controls reduce the potential for infection of cattle and include:
- surveillance for the prompt detection of BSE cases in cattle through the requirement to test certain categories of animal;
- animal feed controls; and
- controls on SRM.
1.25 The key food safety control is the removal of SRM. In countries with CR status (the current status in Scotland), SRM must be sent for destruction by incineration or by rendering followed by incineration. An outline of the current controls in Scotland is attached at Annex A.
Question 3
Do you have any comments to make from a public health perspective in relation to the relaxation of SRM-related controls?
Question 4
Do you have any comments from a consumer perspective?
What would change if Scotland achieved BSE NR status?
BSE Controls
1.26 If Scotland moved from CR to NR status, BSE testing and feed and food safety arrangements for cattle would not change, unless an OIE risk assessment indicated that this was appropriate, and then only in accordance with Regulation ( EC) No. 999/2001. In the event of moving from CR to NR status, feed controls, surveillance etc. would continue to be carried out in accordance with the EU requirements.
1.27 The necessary official controls, therefore, would remain in place to deal with any residual risks associated with occasional cases of BSE. It is anticipated that existing food labelling rules, which are set out in legislation, would be unaffected and would continue to apply.
Benefits
1.28 Industry have argued that BSE NR status would contribute positively to Scotland's global image which will offer commercial benefits in terms of gaining entry into new markets.
1.29 Industry also take the view that there would be a reduction in the volumes of bovine tissues designated as SRM going for destruction which would reduce disposal costs for the red meat sector, and enable them to exploit new revenue streams.
Risks
1.30 The sporadic occurrence of BSE cases, means NR status could be lost as a result of a single case, thereby potentially generating considerable negative publicity.
1.31 There may also be implications for exports to non- EU countries which have agreed terms to trade based on a NR status basis. Should NR status be lost, there would likely be a requirement to renegotiate export conditions and this may lead to the loss of some important markets which were gained as a result of obtaining NR status.
1.32 There is also the risk that, with the reduction in the quantity of SRM for disposal, the rendering industry may be adversely affected, with the possibility that the rendering capacity in Scotland might reduce. Reduction in Scottish rendering capacity would result in reduced competition for material for disposal, with possible implications for increased disposal costs. This could impact on the disposal of fallen stock. The Scottish Government would have concerns if capacity in the fallen stock rendering industry was reduced, and especially in the context of an epizootic disease situation such as foot and mouth disease.
1.33 The implications of BSE NR Status are explored in more detail in Chapter 2.
Contact
Email: Ian Cox, BSEConsultation@gov.scot
There is a problem
Thanks for your feedback