Counselling and advice on medicines and appliances in community pharmacy practice
Web publication of guidelines produced in hard copy in March 1996 for pharmacists working in NHSScotland.
Annex A4: Clinical Audit
1. What is Clinical Audit?
Clinical audit is a process of systematic, critical analysis used by health care professionals to evaluate and improve patient care.
All pharmacists should view clinical audit as an opportunity to improve standards of care and to further their own continuing professional development.
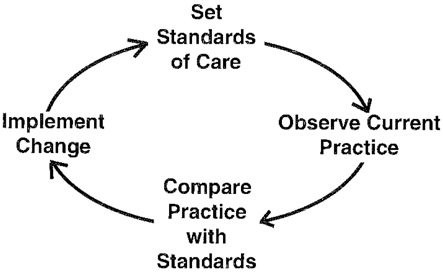
The approach should be non-threatening, educational and professionally led. The audit process is often represented as an audit cycle (Figure 1).
Figure 1: The Audit Cycle
2. Self Audit
2.1. Setting standards
This can involve any or all of the following methods; peer consensus, national guidelines, literature review, previous experience and best guess. Local circumstances can be taken into account when deciding on a desirable level of care. Agreeing standards is integral to audit: it ensures clear objectives and is a valuable educational experience.
2.2 Observing current practice
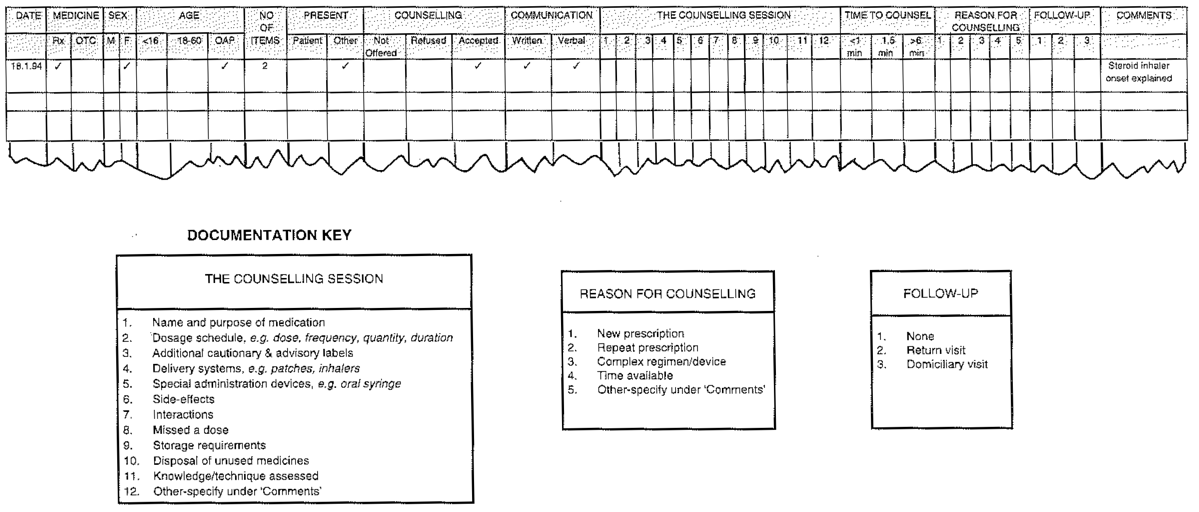
Methods of measuring current practice include survey by questionnaire or interview, direct observation, tally counts and recording practice details. Data collection should be kept as simple as possible, and existing documentation used where appropriate, e.g. patient medication records could indicate when a patient was last advised on correct inhaler technique. An example of data collection form which could be used to audit counselling services is given in Figure 2.
2.3 Comparing practice standards
This identifies those areas of care requiring change. An audit of counselling services may reveal that the proportion of patients receiving instruction on inhaler use is less than expected. Asking why this is so and what can be done may suggest a need to introduce new work practices in to the pharmacy to increase the numbers of patients offered instruction. Alternatively, the audit findings may be used to set realistic standards.
2.4 Implementing change
For audit to be successful it is important to act on the results. Readiness to change is fundamental. Planned changes may be incorporated into an action plan with an agreed timetable. To ensure effective and lasting change it is important to involve, at an early stage, all individuals whose
co-operation is desirable.
The full benefits from audit will only be realised if the audit is repeated on a regular basis, changes are made where appropriate and their impact evaluated.
3. Research and Development in Partnership with Audit
Research, audit and service development are mutually supportive activities with a degree of overlap.
The following may help to distinguish these activities as they relate to patient care:
| Research |
- What is best care? |
| Audit |
- Is the best care being provided? |
| Development |
- How could this service be improved? |
Figure 2: Example of Audit Documentation
Clinical research aims to establish what is the best clinical practice and this information then feeds into the standard setting phase of the audit cycle. Audit examines if best clinical practice is actually being provided. Any deficiencies or problems highlighted may have research implications.
Audit, particularly the evaluation and monitoring stages, also has a valuable contribution to make to the processes of service development. By providing information on current practice, audit can justify the need for service changes. Alternatively, after a service development has been introduced, audit can monitor the impact of the change.
4. Support Structure for Audit
The initial approach of SODoH to establish uniprofessional audit was followed in 1993 by the development of multiprofessional collaborative audit. An audit framework exists at national and local level to promote both professional and clinical audit.
At national level The Clinical Resource and Audit Group ( CRAG), a division of the National Health Service in Scotland Management Executive, exists to promote and support audit as does the more recently established Scottish Clinical Audit Resource Centre (SCARC) in Glasgow. Within each Health Board, Area Clinical Audit Committees exist primarily to develop area audit strategy and facilitate audit as a form of continuing education. Although the current aim is to progress to multiprofessional audit, a number of the uniprofessional Pharmaceutical Audit Committees have been retained.
The importance of audit has been recognised by SCPPE through the provision of both national and local direct learning courses. The distance learning package 'Moving to Audit : an educational package for pharmacists' provides an alternative resource for pharmacists unable to attend direct learning courses.
A natural progression from introductory courses is the formation of local audit groups, facilitated by co-ordinators. Audit toolkits are being developed to offer practical guidance to community pharmacists wishing to participate in audit. These packages contain all the necessary documentation and reference material needed to undertake a simple audit. The national pharmaceutical audit facilitators have prepared a package on "Patient Counselling for Prescribed Medicines" for use by community pharmacists.
Contact
There is a problem
Thanks for your feedback