The Penrose Inquiry recommendation
Short-life working group report on the Penrose Inquiry recommendation on infection of people with Hepatitis C from NHS treatment.
6 Response to each of the Terms of Reference of the Short-Life Working Group
6.1 To assess the extent of the problem - i.e. estimated numbers of living HCV-infected individuals who acquired their infection through blood transfusion in the UK pre-1991 and who remain undiagnosed.
6.1.1 Number of people who received a blood transfusion pre-September 1991 and are still alive
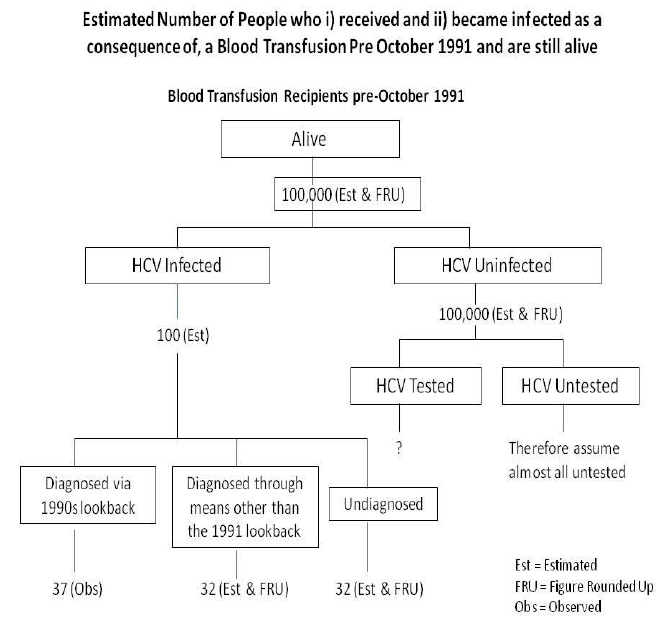
The Scottish National Blood Transfusion Service ( SNBTS) estimates that 93,600 people who received a blood transfusion before-September 1991 were still alive in 2015. This figure is a best estimate, based on incomplete data; it is possible that an error of 10 - 20% in either direction exists (See Appendix 2 for detail). The estimate is based on (i) an estimate of the proportion of Scotland's population transfused, based on SNBTS data, and (ii) an estimate of the life expectancy of people transfused at the time of transfusion, based on General Registry Office life expectancy data for the general population. To ensure that any potential for underestimating the scale of the problem was minimised the figure of 93,600 was rounded up to 100,000.
6.1.2 Number of people who received a blood transfusion pre-September 1991, are still alive but have not been tested for HCV.
It is not possible to estimate, with any degree of accuracy, the proportion of the 100,000 people who received a blood transfusion in Scotland prior to September 1991 and who are still alive and have been tested for HCV. It is likely that a small proportion of these people will no longer be living in Scotland, but this is likely to be offset by people from, in particular, the rest of the UK now living in Scotland. The 2007 SIGN (Scottish Intercollegiate Guideline Network) Guidelines on the management of Hepatitis C recommend that people who had a blood transfusion pre-1991 should be offered an HCV test. However, data from surveys of HCV testing practice among general practitioners in Scotland in 2007 and 2013 indicate that few i) determine if a patient received a blood transfusion pre-September 1991 and, thus, ii) offer an HCV to such individuals. It is uncertain why such practice does not happen to any appreciable extent but it may be partly because the (correct) perception among general practitioners is that the prevalence of infection among the transfused population is extremely low. General Practitioners have not been resourced to perform a significant case finding exercise for these people.
In the absence of evidence indicating what proportion of the 100,000 have been HCV tested the SLWG considers it reasonable to assume - again, for the purposes of avoiding any underestimation of the size of the task - that almost all have not been tested. Accordingly, this assumption would correspond to an average of 17 from the average patient list (1,142) of a general practitioner.
6.1.3 Number of people who received a blood transfusion pre-September
1991, were infected with HCV and are still alive. ( Figure 1)
How many of the estimated 100,000 people who received a blood transfusion in Scotland prior to September 1991 and are still alive were infected with HCV as a consequence of blood transfusion? While Scotland does have a database of all HCV antibody positive people diagnosed in Scotland and a database of nearly all HCV-infected people in specialist care, available information on how people became infected is often suboptimal. Where a history of injecting drug use is recorded, one can be fairly confident that this behaviour was the means through which HCV was acquired. Where blood transfusion is recorded as the risk, in most instances no validation has been undertaken to ascertain if a transfusion was received, where and when it was received, and whether or not the transfused blood was sourced from an HCV RNA positive donor; this is why, for a few hundred living HCV-infected individuals in Scotland, "blood transfusion" is recorded as a possible risk factor.
Accordingly the number of infected individuals is very difficult to estimate; however, we know that the proportion of people receiving a blood transfusion during January - September 1991 who acquired HCV was approximately 1 in 1500; this proportion is based on the observation that 159 of 180,000 (0.088%) donors tested for HCV antibodies by SNBTS during September 91 - February 92 were antibody positive and that an estimated 119 of 180,000 were viraemic on the assumption that 25% of antibody positive individuals would have spontaneously cleared their HCV. It is unlikely that, for any year prior to 1991, the proportion was higher than 1 in 1000 - taking into account both the HCV preventing impact (among donors) of implementing, in 1984, the exclusion of high risk individuals for blood donation and the HCV promoting impact (among donors) of the rapidly increasing prevalence of HCV in the population of Scotland throughout the 1980s. If a 1 in 1000 proportion (i.e. worst case scenario) is assumed, then approximately 100 of the 100,000 individuals transfused prior to September 1991 and still alive are infected.
It is acknowledged that considerable uncertainty surrounding this estimate exists. As at November 2015, 344 living people from Scotland, still alive, had received a payment from the Skipton Fund because it was considered that on the balance of probabilities they had acquired HCV infection through a blood transfusion pre-September 1991.
Figure 1
6.1.4 Number of people who received a blood transfusion pre-September 1991, were infected with HCV and are still alive and have not been tested.
The proportion of the estimated 100 who had been HCV tested and diagnosed will bear no relation to the proportion of the uninfected people having been tested because of the much greater likelihood of an infected, compared to an uninfected, person being tested. This is because of a) the, albeit not fully comprehensive, lookback exercise of the mid 1990s which identified 85 individuals alive at that time who definitely were infected as a consequence of blood transfusion in Scotland pre-September 1991, b) the practice of HCV testing of people in hospital or in the community who have abnormal liver function tests - in particular a raised Alanine Amino transferase ( ALT) enzyme (note that the 2006/7 SIGN Guidelines on the management of Hepatitis C recommended that anyone with an otherwise unexplained raised ALT should be HCV tested), c) the HCV testing of people who present clinically with symptoms and signs suggestive of liver disease and, d) the HCV testing of the more at risk transfusion recipients with conditions requiring multiple transfusions; such individuals, because of their underlying conditions may be more likely to be no longer alive but some, especially younger people, will still be living.
So, even if only a small proportion of the 100,000 people estimated to have had a blood transfusion pre-September 1991 and still alive, have been tested for HCV, a much larger proportion of those infected as a consequence of transfusion pre-1991 will have been diagnosed already.
If we assume 100 infected people, having acquired their HCV infection in Scotland as a result of blood transfusion pre-1991, are still alive in Scotland, it is likely that well over 50% of these will have been diagnosed; this is because the number of infected people diagnosed through the lookback exercise, and still alive in 2014, is 37 and an unknown number of others will have been diagnosed as a consequence of reasons b, c and d as above.
Accordingly, the number of undiagnosed HCV-infected people, having acquired their HCV in Scotland as a result of blood transfusion pre-1991, is within the range of 0 and 63 (where 0 assumes everyone is diagnosed and 63 assumes no cases over and above the 37 lookback ones have been diagnosed). As explained above, a proportion of the non-lookback cases will have been diagnosed. If we assume half of the 63 have been diagnosed, the figure is 32 (figure rounded up); this means that in the context of an estimated 100,000 untested individuals having had a blood transfusion pre-1991, the ratio of HCV infected to HCV uninfected individuals, not yet tested for HCV, is about 1 in 3000 (i.e. 32/100,000). This figure of 32 means that only 1 in 50 individual GPs will have one of these infected, undiagnosed people on their list.
6.1.5 Number of people who received a blood transfusion pre-September 1991, were infected with HCV and are still alive, have not been tested and have, or are likely to progress to severe liver disease.
Of the 32, the proportions of those with no, mild, moderate and severe disease are unknown; however it is possible that the majority of this undiagnosed cohort, currently, are more likely to belong to the milder end of the spectrum because of the greater chance of being diagnosed if one is symptomatic; that said, people with severe disease can remain undiagnosed because of lack of symptoms or failure to be tested despite symptoms.
6.2 Number of people who received plasma products pre-May 1987, were infected with HCV and are still alive but have not been tested.
Virtually all such individuals have been tested. A total of 478 persons with a bleeding disorder were identified as HCV infected; of these, 193 were alive as at March 2015. Group Members representing blood factor recipients indicated that a small number of people, with mild blood factor conditions, who only received occasional treatment, need to be traced and contacted; this work is being undertaken by Dr Campbell Tait in association with Health Protection Scotland and the Information and Statistics divisions of Public Health and Intelligence, National Services Scotland. The work involves securing approval to link identities from a list of individuals on a blood factor deficiency database held by clinicians, with a register of people currently assigned to a general practitioner in Scotland, and then making contact with "linked" people about Hepatitis C testing. For those individuals not known to be dead and not identified as being assigned to a general practitioner in Scotland, additional efforts to trace them will be undertaken.
6.3 What impact did the media response to the publication of the Penrose Inquiry have on HCV testing uptake and HCV positive yield in the relevant population? ( Appendix 3)
The Penrose Inquiry Report was published on the 25 th March 2015. That day, apologies to all those affected were made in UK and Scotland Parliaments by the Prime Minister, David Cameron and the Cabinet Secretary for Health and Wellbeing, Shona Robison, respectively. Additionally, a number of infected individuals and family members gave interviews about their predicament; in the context of what is a stigmatised condition, their courage should be acknowledged and greatly appreciated. The Report's conclusions and its only recommendation, regarding HCV testing, were the lead stories in most newspaper and TV news programmes. It is acknowledged that an appreciable minority of people do not access information through such media.
A letter from Scotland's Chief Medical Officer was sent, on 27 th March, to all GPs in Scotland; it stated that "every effort should continue to be made to offer testing to anyone who may have been exposed to Hepatitis C via infected blood".
An analysis of HCV test data from three of Scotland's Health Boards: Lothian, Tayside and Grampian, was undertaken to examine the impact of the media response. During the 12 weeks before March 25, seven HCV tests, for which there was an indication (in accompanying blood sample report forms) of blood transfusion risk, were performed. During the 12 weeks after March 25, 400 HCV tests, for which there was a mention of blood transfusion and/or the Penrose Inquiry, were performed; females over the age of 50 were particularly represented. This figure is an underestimate as in many instances clinical details on request forms were either unavailable or may not have indicated the reason for the test being done. Less than five of the 400 HCV tests performed generated an HCV antibody positive result. No validation was undertaken to ascertain if the individual(s) had a blood transfusion in Scotland pre-1991 and, if so, whether or not the infection was acquired through blood transfusion. Extrapolating the 400 for Lothian, Tayside and Grampian to all Scotland would generate a figure of approximately 1,000. Note that figures here have been rounded up.
6.4 To review past and current interventions to promote the diagnosis of HCV-infected individuals who acquired their infection through blood transfusion in Scotland pre-1991.
- In 1995, a look back exercise was undertaken to identify transfusion recipients of blood from HCV antibody positive donors diagnosed following the introduction of the HCV antibody test in 1991; this initiative led to the identification of some but not all individuals infected with HCV as a consequence of transfusion.
- In 1999/2000, a Scottish Needs Assessment Report on Hepatitis C listed a range of population groups who should be offered an HCV test; the list included people who had received a blood transfusion pre-1991. In 2002, the Scottish Government distributed an educational Hepatitis C pack to general practitioners throughout the country; the list of people who should be offered a test, as above, was incorporated into the pack's literature.
- In 2006/07, SIGN Guidelines on the management of Hepatitis C were published; these included a list of people who should be offered an HCV test and within this list was "people who had received a blood transfusion pre-September 1991".
- In 2007, the Scottish National Party published, in its Manifesto, a commitment to hold an inquiry into "the infection of people with Hepatitis C from NHS Treatment"; in April 2008, the Cabinet Secretary for Health and Wellbeing made a statement to the Scottish Parliament announcing the establishment of the promised Inquiry. Once established, the Inquiry issued a public call for evidence inviting all interested parties to contact the Inquiry to provide statements. These announcements, accompanied by news stories and adverts, drew public attention to the issue.
- In 2009, within the context of the Scottish Government's Hepatitis C Action Plan, the Chief Medical Officer for Scotland sent a letter to GPs, outlining the at-risk groups (including the blood transfusion one) who should be offered an HCV test; the SIGN Guidelines, as above, were referred to.
- In 2010, the Scottish Government sponsored a national poster campaign involving, in particular, the placement of such materials on the sides of bus shelters.
- In recent years, the UK Hepatitis C Trust and Hepatitis Scotland have staged major awareness campaigns around, in particular, World Hepatitis Day (July 28 th).
- In 2015, the publication of the Penrose Inquiry Report was a leading news story - one which generated, arguably, the greatest intensity ever experienced in Scotland of awareness-raising around the issue of Hepatitis C risk and blood transfusion pre-September 1991.
- Note that numerous local awareness-raising initiatives have been performed by NHS Boards over the years; most, however, have focussed attention on people who have injected drugs and/or people who have originated from high HCV prevalence countries.
- See 6.1.2 for an assessment of the impact of such transfusion specific guidance on HCV testing offer.
6.5 To consider if any further national/centralised action should be taken to identify such individuals in the context of action already taken and the likelihood of appreciable benefit.
The following appraisal of potential actions to identify infected, undiagnosed persons, was undertaken; each was assessed in terms of its practicality, effectiveness and cost.
6.5.1 Potential action: The retrospective testing of blood donor specimens
Detail: see Appendix 4
- HCV testing of stored specimens from people who donated blood between 1984 and 1991.
- Following the identification and confirmation of HCV infected components, the tracing of recipients through hospital blood bank and patient records.
- For those who may have received such a donation, a further investigation to determine if they are still alive and, if so, are not already known to be HCV infected.
- Finally, the tracing of such individuals, making contact with them and then offering them an HCV test.
Practicality/effectiveness
- An enormous task involving an estimated 2 to 2.5 million specimens and associated paperwork which will need to be searched, tested and reconciled by hand. For comparison SNBTS currently tests around 250,000 samples per annum using automated testing and IT linkage.
- There are many issues with the reliability of the archive related to the integrity of the samples and records:
- Linking tested samples to specific donors, components and the hospitals they were provided to is likely to be difficult and may not be possible in many cases due to the variety of paper-based and early IT systems used at the time.
- The samples were collected in a variety of different formats in different centres and have been stored under a variety of conditions over a long period of time. In addition, assay methodologies have not been optimised for this nature. Therefore the quality of the samples and the integrity of the results cannot be guaranteed.
- Hospital record retention does not extend back to the 1984-1991 period and it would be unlikely that patients who received specific components could be identified (or traced).
- An effort would also need to be made to trace test positive donors to inform them and carry out confirmatory testing. Most of these people will no longer be blood donors and may have moved or themselves be deceased.
- This would be a major exercise, incurring significant costs and requiring 6 - 7 or more years to complete.
- A break in the chain of traceability in any of the above areas would negate the benefit of testing. In reality there are significant weaknesses in all areas. The likelihood of being able to trace significant numbers of infected patients through this route is small.
Cost
- £8 - 10 million not including the costs of patient tracing.
6.5.2 Potential Action: The interrogation of clinical records to determine blood transfusion history
Detail
- The interrogation of all GP or hospital records of individuals born before 1991 to determine who had received a blood transfusion pre 1991 and were still alive.
Practicality/Effectiveness
- The mostly manual investigation of case notes of all people in Scotland before 1991
- It is likely that many records for the period pre 1991 no longer exist
- Information indicating whether or not someone had been transfused may not have been communicated to the GP
- In the context of an estimated 93,600 living people having received a blood transfusion pre-September 1991 and around 29 of them having undiagnosed HCV infection - effectiveness likely to be extremely low.
Cost
- Probably millions of pounds for administrative/ clerical activity.
6.5.3 Potential Action: Notifying all GP patients about the pre-1991 transfusion risk
Detail
- A letter from GPs to all patients asking them if they had received a blood transfusion pre 1991 or if they had a condition which conveyed a high risk of transfusion.
- Offering those saying "yes" to either of the above, a hepatitis C test
Practicality/Effectiveness
- Essentially writing to all adults in Scotland over the age of 25.
- Very considerable uncertainty about the number of people who might respond to the ask; many might not know or remember having had a transfusion.
- Potential to create unnecessary anxiety amongst the population leading to requests for reassurance from the worried well.
- Probably only a minority of the total number of individuals transfused and not tested before, plus an unknown number of people who thought they might have had a transfusion but did not, would be tested
- Effectiveness probably low.
Cost
- Probably millions taking into account general practice admin costs plus testing ones.
6.5.4 Potential Action: Delivering a targeted awareness campaign focussed on the pre-September 1991 blood transfusion risk and the recent advances in Hepatitis C treatment and care.
Detail
- An initiative run by the Scottish Government in association with Health Scotland, National Services Scotland ( SNBTS and HPS), Hepatitis Scotland and the UK Hepatitis C Trust.
- An initiative involving the health sector and other agencies, using traditional (e.g. leaflets and posters) and non-traditional (e.g. social media) methods.
- Building on the publicity generated through the launch of the Penrose Inquiry report.
Practicality/Effectiveness
- Effectiveness in raising awareness among most of the population of Scotland likely to be high. Effectiveness of identifying the remaining smaller number of HCV infected, undiagnosed individuals uncertain.
Cost
- Hundreds of thousands of pounds for the campaign and the cost of testing those coming forward for a test.
Other Non-Specific Actions Which Might Have an Effect
6.5.5 Potential Action: General population HCV screening
Detail
- Offering, via a letter, an HCV test to all individuals in Scotland over the age of 24.
Practicality/Effectiveness
- Only an unknown proportion would take up the offer.
- The action woul d identify some of the 15,000 undiagnosed HCV infected individuals in Scotland; nearly all would be people who had ever injected drugs.
- In the context of around 30 undiagnosed infected people who acquired their HCV through transfusion pre 1991 - the action is likely to be very ineffective. However, in the context of the rest of the undiagnosed HCV infected population it is likely to be more effective. To achieve much greater effectiveness in relation to identifying people who had injected drugs in the past, such general population screening would be best confined to certain geographical areas where injecting drug use is and has been highly prevalent.
Cost
- Tens of millions of pounds
6.5.6 Potential Action: Reminder to all clinicians to test for hepatitis C if someone has an otherwise unexplained raised ALT
Detail
- A letter from the CMO to all hospital clinicians and general practitioners to remind them of the 2006/7 SIGN guidelines on testing people who have an otherwise unexplained raised Alanine Amino transferase ( ALT).
- The letter would also ask the clinicians to test for HCV even if the raised ALT might be explained by excessive alcohol consumption ( EAC); this is because of the strong relationship between EAC and injecting drug use (and thus hepatitis C infection).
- It is uncertain what proportion of individuals with a raised ALT, as above, are tested for hepatitis C but preliminary information indicates that clinical adherence to the guidance around the ALT criterion is suboptimal.
- Research is being undertaken in 2016 to assess clinical adherence to such testing and, thus, whether or not a CMO letter as above would be warranted.
Practicality/Effectiveness
- Not everyone has their liver function tested and not everyone with advancing HCV disease has a raised ALT at the time the test is performed
- In the context of the 15,000 undiagnosed infected individuals in Scotland, increasing awareness around ALT-based hepatitis C testing might be effective; however, it is uncertain what impact a CMO letter might have on clinical practice
- Nevertheless, current guidelines state that HCV testing of individuals with an otherwise unexplained raised ALT should be undertaken as part of routine clinical investigation.
Cost
- Any increase in HCV testing to identify the cause of liver damage, as indicated by a raised ALT, should not be judged in terms of cost (and cost effectiveness) in the same way that a "screening" intervention should be. The difference between clinical investigation and screening is a critical one.
6.6 SLWG: Recommendations
The SLWG fully acknowledges the importance of ensuring that anyone still alive and infected with Hepatitis C as a consequence of blood transfusion pre-September 1991 should be given the best possible chance of taking advantage of the new advances in treatments which are safe, easy to administer and highly effective. Appropriate opportunities to access counselling and psychological therapies should also be available for the newly diagnosed, as is already good practice. Taking account of the Inquiry's recommendation that any steps to offer an HCV test to such individuals should be "reasonable", the SLWG also recognised the need to ensure that any new intervention(s) are optimally cost-effective and proportionate in the context of the scale of the problem. Accordingly, the proposed Actions 6.5.1, 6.5.2, 6.5.3 and 6.5.5, as above, were regarded by the SLWG as inappropriate because of their high cost (millions of GBP) and very likely low effectiveness.
Accordingly, the SLWG unanimously agreed on the following:
- Delivering a targeted awareness campaign focussed solely on individuals who received a blood transfusion pre-September 1991
This awareness campaign should aim to reach all targeted individuals through the use of (e.g. leaflets and posters) and more modern (e.g. social media) approaches. Such approaches recognise that an appreciable minority of people do not access information from more traditional sources. The details surrounding the design and implementation of the campaign would be worked on following any such Scottish Government approval. The SLWG agreed that any such campaign should be evaluated to determine its impact.
- The identification and written offer of an HCV test to a group (up to 71) of plasma product factor recipients who are as yet not known to have been HCV tested.
- A Chief Medical Officer letter should be sent to all clinicians in Scotland to remind them of certain risk factors (including pre-September 1991 blood transfusion and injecting drug use) and clinical (including otherwise unexplained Alanine Aminotransferase ( ALT) liver enzyme level) indicators for HCV infection and making them aware of the recent advances in therapy and thus the benefits of HCV testing.
Contact
Email: Robert Girvan, robert.girvan@gov.scot
There is a problem
Thanks for your feedback